A spark arched between them and with it a cloud of carbon gas puffed into existence, vaporizing the tip of the anode rod. As the carbon-laden air settled on the chamber walls it formed a thin layer of black soot, within it a strange new material appeared. Tiny single-layer straws of carbon. Sumio Iijima had just created carbon nanotubes.

Laboratory testing of these mysterious little tubes in the following years would reveal that these nanometer-wide hexagonal lattices of carbon had the strongest tensile strength known to man, and this was just As one of the many incredible material properties they displayed. Carbon nanotubes are light, conductive, and biocompatible.
It soon became clear that the carbon nanotube had the potential to be the building block of futuristic new technologies. The most efficient computers, transformative medical devices, synthetic muscles, or perhaps the most ambitious of all, space elevators, the dream of countless sci-fi authors, Carbon nanotubes has promised to be the catalyst for the next revolution in technology.
But, putting this revolutionary material to work will not be easy. It turns out that building a fiber, that is actually a single molecule, of any significant length is incredibly difficult. To understand this fascinating molecule, let's dive into the chemical makeup of carbon nanotubes.
Carbon is a very familiar element. It’s in everything we eat, sleep on and step over. It is the element that holds our DNA together. It forms the carbohydrates, proteins, and lipids that we depend on to build and fuel our bodies.
It’s the basis of life as we know it. Its ubiquity in our lives is a result of its versatility. It’s chemical properties allow it to take many different shapes, each impacting its material properties in diverse and unique ways. To understand this we need to understand the basic models of how we visualize electron orbits around the nucleus of an atom.
To start we have the simplified Bohr model, which separates the electrons into shells. The first shell can contain 2 electrons, while the next shell can hold 8. An atom wants to fill each shell to be stable. Let’s take an atom of carbon, which has 6 electrons, to see how this plays out.
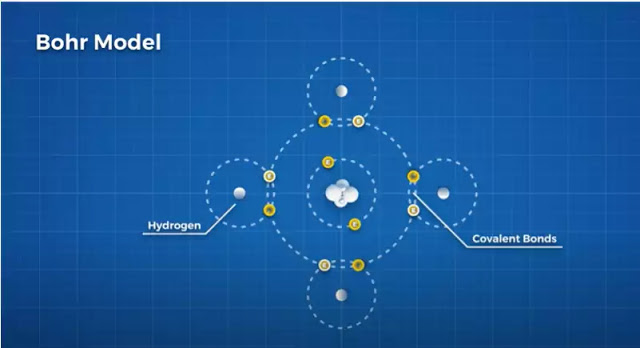
First, we fill the first shell with it’s2 electrons, then we have 4 electrons left to fill the next shell, leaving 4 open positions in its outer shell The 4 open positions mean that carbon willingly interacts with many other elements as well as itself. Often by sharing electrons in a special type of bond, called a covalent bond.
This versatility allows carbon to create many different kinds of molecules. Take hydrocarbons. Hydrogen has 1 electron and seeks 1 electron to fill its inner shell. So, carbon likes to form 4 covalent bonds with 4 hydrogen atoms to form a stable 8 electron outer shell while helping hydrogen form a stable 2 electron shell.
This is methane, an incredibly common molecule that is the main ingredient in natural gas fuels. This is just one arrangement carbon can take. Hydrocarbons take a huge range of shapes and configurations, but what we are interested in is how carbon bonds to itself, but this simplified Bohr model doesn’t give us an understanding of how carbon to carbon bond take radically different shapes.
We need to dive a little deeper before we can understand the magic of carbon nanotubes. Electrons don’t travel in neat 2D circular orbits as the Bohr model would suggest, in fact, we can’t even know the position and speed of an electron.
Instead, we can make predictions about electrons' general locations in 3D space. We call these orbitals, and they are regions where we have about a 90% certainty that an electron is located somewhere within that region. This can get pretty complicated, but we just need to concern ourselves with two types for now. S and P orbitals.
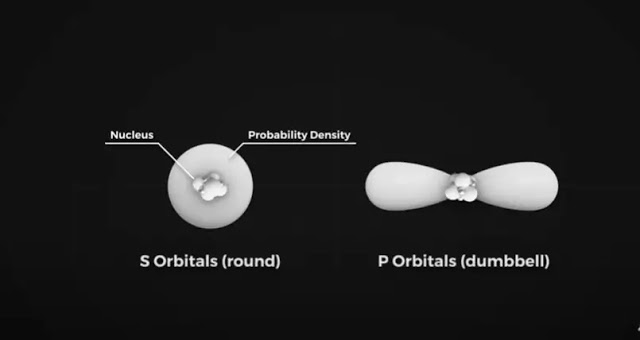
S orbitals are spherical in shape with the nucleus of the atom at their center. P orbitals are often called dumbbell-shaped, but we don’t know what gym these nerds are going to, because I have never seen a dumbbell like this.
It’s more like a figure of 8 shapes like the infinity symbol. In the ground state, electrons will occupy the lowest energy orbitals first, which in this case is the 1S orbital. It can hold two electrons.
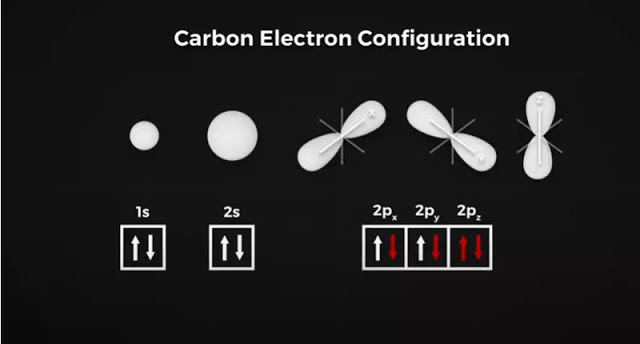
Next, we have the 2S orbital, which is a larger sphere and can also hold 2 electrons. Then we have our three P orbitals, one aligned along the X, Y, and Z-axis, each capable of holding 2 electrons. Carbon in its ground state has the 1S and2S orbitals filled, with one electron in the Px orbital and one in the Py orbital. To be stable, Carbon wants to fill these three p orbitals with 2 electrons each.
Now this where things get a little funky and confusing, and it will be on your final exam. Carbon can bond to itself in different ways that affect these orbital shapes. Take diamonds. To fill these orbitals, carbon bonds with4 neighboring carbon atoms. To do this it promotes one electron from its 2S orbital into the empty Pz orbital.
This Pz orbital is higher energy than the 2S orbital, and the electron doesn’t want to stay there, so the carbon atom takes on new hybrid orbital shapes to compensate. This is called sp3 hybridization, which is a mixture of S and P orbital shapes and looks something like this.
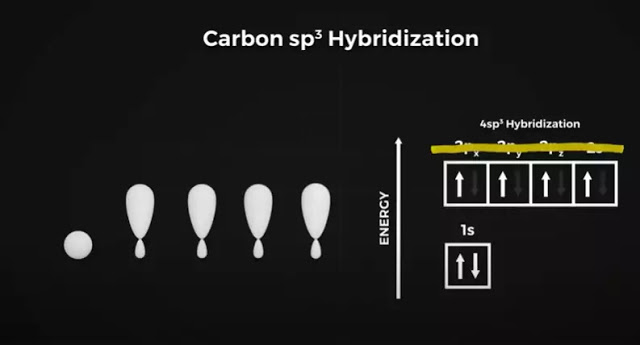
Where one side of the figure of 8 expands while the other contracts. The 2S and 3 P orbitals are transformed into these new SP3 orbital shapes. They repel each other equally in this 3D space to form this four-lobed tetrahedral shape with 109.5 degrees between each lobe.
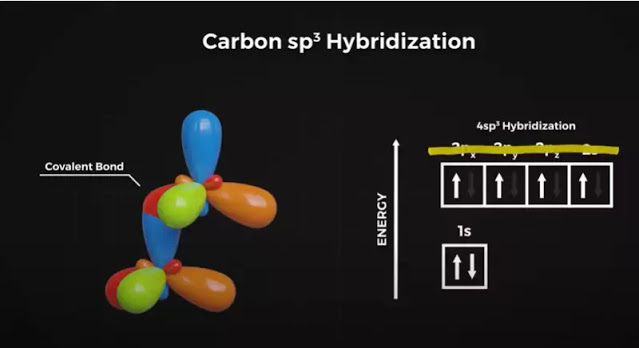
Covalent bonds now form between the carbon molecules where these orbital lobes overlap head-on in what’s called a sigma bond.

This creates a repeating structure like above and it’s this rigid framework of carbon atoms that makes diamond extremely hard. Now, what’s fascinating to me, is that you can take the same carbon atoms and now form graphite, a material so soft that we use it as pencil lead and as a lubricant.
How does that work? Here a different hybridization occurs. Once again 1 electron from the 2S orbitalis is promoted into the Pz orbital, but this time the S orbital hybridizes with only 2of the P orbitals, giving us the name SP2 hybridization.
This gives us three SP hybrid orbitals and 1 regular P orbitals. This new arrangement causes the orbitals to take a new shape, with the 3 SP orbitals arranging themselves in a flat plane separated by 120degrees, with the P orbital perpendicular to them.
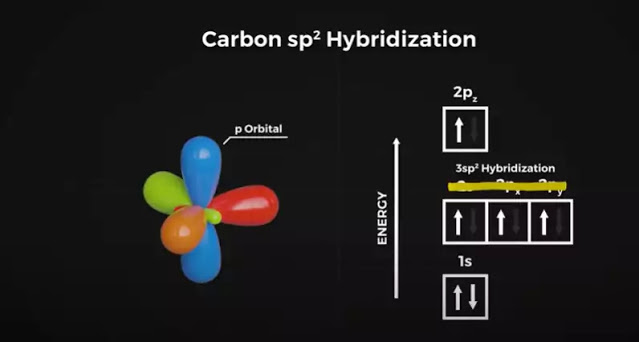
Now, when the carbon atoms combine, the heads of the SP orbitals overlap once again to form this flat hexagonal shape. A hexagon pattern is naturally a very strong and energy-efficient shape. For example, bees don’t intentionally build honeycombs in hexagons. They form as a result of the warm bee bodies melting the wax and the triple junction hardens in the strongest formation.
The shape is frequently used in aerospace applications where high strength and low weight is a priority. These SP2 bonds are stronger than SP3 bonds because they have a higher s character. This sounds complicated, but all it means is that they are more like S orbitals than a P orbital.
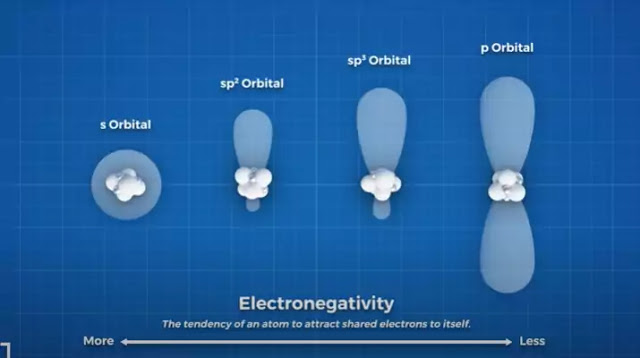
Because there are 3 SP bonds, they have a 33% S character, whereas SP3 orbitals have 4 SP bonds giving them 25% S character. S orbitals are closer to the nucleus, making SP2 bonds shorter and more electronegative than SP3 bonds, and thus stronger.
This hexagonal structure and strong bonds make graphene exceedingly strong. Laboratory testing of graphene using atomic force microscopes has shown graphene has a young's modulus of 0.5 TPa and ultimate tensile strength of 130 gigapascals.
So strong that if we could somehow create a large perfect sheep of graphene, which we can’t, we could build an invisible single atom deep hammock that could support the weight of a cat.
Imagine the number of cats we could confuse. That’s the world I want to live in. That’s an entertaining, but not terribly useful application, but graphene is a very common material and the form we are used to, graphite, is not strong.
This hexagonal shape itself is extremely strong, but because graphite forms these single-atom layer sheets with only weak van der Waal forces holding them together, the sheets can easily slide over each other, which is the reason graphite is so soft.
Now what is interesting is that carbon nanotubes take the same repeating hexagonal structure as graphite. The ends of the sheets are simply looping and connect with themselves to form a tube, and this structure is what gives carbon nanotubes their incredible strength. Researchers found that single-walled nanotubes have strength similar to that of graphite, about 130 Gigapascals.
For the non-engineers in the crowd, let me rephrase that. It’s a lot. About 100 times greater than steel, and to boot, it’s vastly lighter. If this material could be feasibly manufactured into a single extremely long fiber, it could potentially open up entirely new design possibilities.
So where are there space elevators? Here lies the difficulty. Manufacturing carbon nanotubes. Carbon nanotube's strength relies on creating a continuous perfect lattice of carbon atoms in a long tube, and that process is not something we have yet developed.
So how can we create carbon nanotubes? Things have changed a bit since the days of Sumio Ijima’s first discovery. The most promising method for industrial-scale production of high purity carbon nanotubes is chemical vapor deposition.
In this manufacturing method, a precursor gas containing carbon, like methane (CH4) is introduced into a vacuum chamber and heated. As the heat increases inside the chamber, the bonds between the carbon and hydrogen atoms begin to decompose.
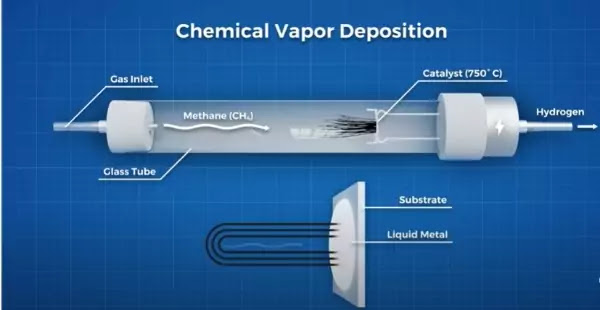
The carbon then diffuses into a melted metal catalyst substrate. This then becomes a metal-carbon solution, which eventually becomes supersaturated with carbon. At this point, the carbon starts to precipitate out and form carbon nanotubes.
While the hydrogen bi-product is vented out of the chamber to avoid an explosion. Our research has focused on increasing the length of these nanotubes while not sacrificing their structure, yield, or quantity. While some labs have gotten individual tubes as long as 50 cm it’s been a struggle to get larger bundles of tubes, which are called forests, to a length greater than 2cm.
This is because the catalyst is guaranteed to deactivate at some point during the growth process, terminating the growth of the nanotube. The key to growing longer nanotubes is minimizing the probability of the catalyst deactivation.
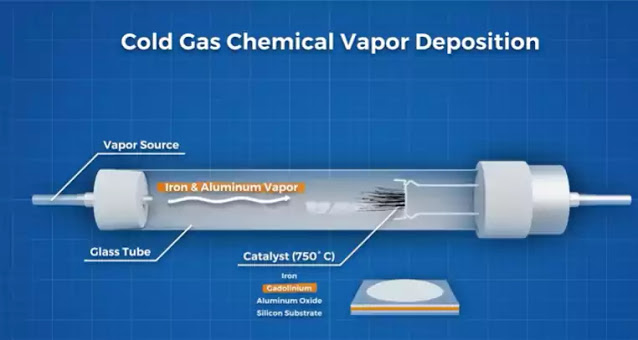
In 2020, a research team based in Japan managed to grow a forest over 15 cms in length, 7 times longer than anyone else, using a new method of chemical vapor deposition that managed to keep the catalyst active for 26 hours.
They did this by adding a layer of gadolinium to a conventional iron-aluminum oxide catalyst coated onto a silicon substrate. Then using a lower chamber temperature, small concentrations of iron and aluminum vapor were added into the chamber. These factors combined managed to keep the iron-aluminum oxide catalyst active for much longer.
This method is a major leap forward that could allow carbon nanotube products to begin entering the market, but we are still a long way from a space elevator. Most products today call for the fibers to be woven together to form a textile-like yarn.
One study found wove together 1 mm long nanotubes and into a yarn and then impregnated that with an epoxy resin to form a composite material, which had a pretty good tensile strength of 1.6 Gigapascals.
Beating aluminum in its strength to weight capabilities, but below a traditional carbon fiber composite. However, these new longer nanotubes may give us stronger woven fibers in the future. It’s important to remember that carbon nanotubes aren't just strong.
Their most exciting new term applications will come about as a result of their other material properties. Like their conductive abilities. Like graphite, nanotubes are highly conductive, because each carbon atom is only bonded to 3 other carbon atoms, each atom has 1 free valence electron available for electrical conduction.
Making carbon nanotubes excellent conductors. The conducting core of cables that make up our overhead grid lines is typically made from aluminum. Even though aluminum is a poorer conductor than copper, and thus causes a greater loss in power over the lines. It’s used because it’s cheaper and lighter.
Allowing support structures for overhead lines to be spaced further apart. Individual nanotubes are orders of magnitude more conductive than copper, but creating a yarn of nanotubes that could match copper has been a challenge.
Electrons move through individual nanotubes very efficiently, but when the tube comes to an end the current meets resistance when jumping to a neighboring tube. So these longer tubes developed last year are opening doors to conductors that are vastly lighter than aluminum and more conductive than copper.
These could be used for grid connections, allowing our power lines to be stretch further without supports and minimize the loss of power to heat resistance, but for now, the price of nanotubes likely shuts that door.
Instead, we could see these wires being used in super lightweight aircraft or cars. They are even being investigated as a means of helping composite structure planes, like the 787, survive lightning strikes.
The 787 is primarily composed of carbon and glass fiber reinforced plastics, but because they do not conduct electricity, the plane has additional conductive structures added to protect it from lightning strikes, like a thin copper mesh.

This mesh adds weight that increases fuel consumption. This could be drastically reduced by including a carbon nanotube mesh on the surface of the composite instead. Nanotubes are quite elastic. Capable of stretching to 18% of their original length and returning to their original shape after.
This could allow conductive carbon fiber wires to be incorporated into wearable technology. The carbon fiber threads can even be treated as a normal thread and sewn into a fabric using a sewing machine. Perhaps the most exciting application is in biomedical devices.
Carbon nanotubes are biocompatible. Meaning they are not toxic, nonreactive, and do not elicit an immune response. Combine this with their conductivity, flexibility, and strength, nanotubes become extremely attractive as neural interface material.
A large part of Neuralink, Elon Musk’s neural interface companies, efforts have been focusing on creating smaller wires and the machines needed to implant them. Larger, stiffer wires tear into flexible brain tissue over time and cause scar tissue to form around the wire, preventing signals from passing from the neurons to the wires.
Nanotube wiring could be made smaller and more flexible while being accepted by the body. A potentially game-changing material for biomedical implants.
As with every major material innovation, Fromage hardened aluminum ushering in a new age of aviation, to silicon semiconductors opening up an entirely new world for computers, carbon nanotubes have the potential to open the door to design possibilities and technologies we have yet to imagine. New materials radically change how and what we build, and learning more about manufacturing processes for complex machines really gives you insight into how a material like this could change things.
Read the original article on Hire Blogger Designe.
