Recent advances of Alnylam Pharmaceuticals, Inc. in lung delivery technology have laid the groundwork for developing a successful treatment for COVID-19, the disease that is rapidly traveling across the globe, leaving a rising death toll in its wake. The company has turned its focus to the immediate development of this potential treatment in collaboration with Vir Biotechnology Inc. which has particular expertise in the treatment and prevention of infectious diseases. The companies have been closely partnering since 2017, and thus far, succeeded in developing six novel small interfering RNAs (siRNAs) to treat infectious diseases. Their new RNA interference (RNAi) therapeutic for the treatment of chronic hepatitis B is currently being tested in clinical trials.
In their current collaboration, Alnylam’s proprietary lung delivery technology is employed; it is based on siRNAs, which are the molecules that mediate RNA interference, a natural cellular process of gene silencing. In the case of the new coronavirus – also known as SARS-CoV-2, – Alnylam has designed and prepared over 350 siRNAs that can target all the available genomes of SARS-CoV-2 and even its highly conserved regions. These siRNAs will then be screened in in vitro potency assays, and the best candidates of the potent siRNAs will be further evaluated by Vir’s scientists for their in vitro and in vivo antiviral activity.
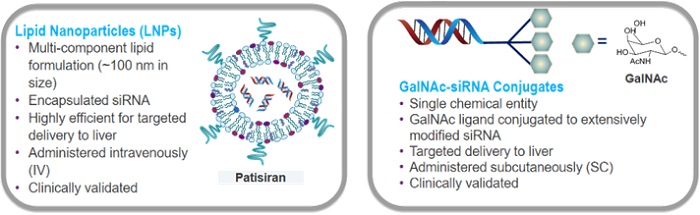
Alnylam utilizes two platforms for the delivery of siRNAs to target tissues, both of which designed based on lipid nanoparticles. In these platforms, the designed siRNAs are firstly functionalized by appropriate linkers. A linker is a cleavable functional group that is specifically determined for the targeted tissue; for instance, a suitable linker for targeting and delivering siRNAs to liver cells – which are rich in esterase – would be ester groups. Functionalized siRNAs are then encapsulated into different types of nanoscale capsules, including lipids (e.g., stable nucleic acid lipid particles), liposomes, cyclodextrins, etc., so as to easily reach and enter the targeted cells. This revolutionary approach is expected to transform the conventional treatment methods of genetic and other serious diseases.
This is a revolutionary approach with the potential to transform the care of patients with genetic and other diseases.
Alnylam platforms for functional siRNA delivery to target tissue.
SNALP is a lipid nanoparticle based siRNA trigger formulation that comprises ionizable lipids, shielding lipids, cholesterol, and endogenous or exogenous targeting ligands such as ApoE lipoprotein. Another platform of siRNA trigger with nanoparticles is GalNAc-siRNA, a liver-targeted triantennary siRNA conjugate used in several drug candidates by Alnylam.
About RNAi
RNAi (RNA interference) is a natural cellular process of gene silencing that represents one of the most promising and rapidly advancing frontiers in biology and drug development today. Its discovery has been heralded as “a major scientific breakthrough that happens once every decade or so,” and was recognized with the award of the 2006 Nobel Prize for Physiology or Medicine. By harnessing the natural biological process of RNAi occurring in our cells, a new class of medicines, known as RNAi therapeutics, is now a reality. Small interfering RNA (siRNA), the molecules that mediate RNAi and comprise Alnylam's RNAi therapeutic platform, function upstream of today’s medicines by potently silencing mRNA that encode for disease-causing proteins, thus preventing them from being made.
About Alnylam
Alnylam is leading the translation of RNA interference into a whole new class of innovative medicines with the potential to transform the lives of people afflicted with rare genetic, cardio-metabolic, hepatic infectious, and central nervous system (CNS)/ocular diseases. Founded in 2002, Alnylam is delivering on a bold vision to turn scientific possibility into reality, with a robust RNAi therapeutics platform. Alnylam has a deep pipeline of investigational medicines, including five product candidates that are in late-stage development.
About Vir Biotechnology
Vir Biotechnology is a clinical-stage immunology company focused on combining immunologic insights with cutting-edge technologies to treat and prevent serious infectious diseases. Vir has assembled four technology platforms that are designed to stimulate and enhance the immune system by exploiting critical observations of natural immune processes. Its current development pipeline consists of five product candidates targeting hepatitis B virus, influenza A, human immunodeficiency virus and tuberculosis.
Sources:
- Vir and Alnylam Expand Collaboration to Advance RNAi Therapeutics for the Treatment of Coronavirus Infection, Including COVID-19
- The Patent Landscape of siRNA Nanoparticle Delivery
- US patent: 195557139
- Platform Advances in RNAi Therapeutics
- Delivery materials for siRNA therapeutics







