Named after Russian mineralogist Lev Perovski, perovskite materials have a crystal-lattice structure of inorganic molecules like that of ceramics, along with organic molecules that are interlaced throughout. Up to now, these organic molecules appeared to only serve a structural function and could not directly contribute to perovskites’ electronic performance.
Led by UCLA, a new study shows that when the organic molecules are designed properly, they not only can maintain the crystal lattice structure, but also contribute to the materials’ electronic properties. This discovery opens up new possibilities to improve the design of materials that will lead to better solar cells and LEDs. The study detailing the research was recently published in Science.
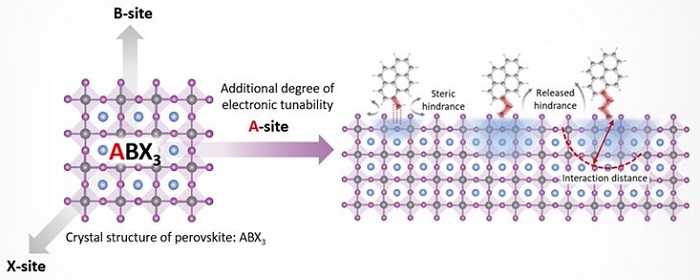
Schematic of perovskite material with organic molecules that can add to its electronic properties.
“This is like finding an old dog that can play new tricks,” said Yang Yang, the Carol and Lawrence E. Tannas Jr. Professor of Engineering at the UCLA Samueli School of Engineering, who is the principal investigator on the research. “In materials science, we look all the way down to the atomic structure of a material for efficient performance. Our postdocs and graduate students didn’t take anything for granted and dug deeper to find a new pathway.”
On its exterior, the positively charged ammonium molecule connected to molecules of pyrene — a quadruple ring of carbon atoms. This molecular design offered additional electronic tunability of perovskites.
“The unique property of perovskites is that they have the advantage of high-performance inorganic semiconductors, as well as easy and low-cost processability of polymers,” said study co-lead author Rui Wang, a UCLA postdoctoral scholar in materials science and engineering. “This newly enhanced perovskite material now offers opportunities for improved design concepts with better efficiency.”
To demonstrate perovskites’ added effectiveness, the team built a photovoltaic (PV) cell prototype with the materials, and then tested it under continuous light for 2,000 hours. The new cell continued to convert light to energy at 85% of its original efficiency. This contrasts with a PV cell made of the same materials, but without the added altered organic molecule, which retained only 60% of its original efficiency.
Read the original article on University of California, Los Angeles (UCLA).