The 2020 Chemistry Nobel Prize-winning technology CRISPR/Cas9 holds great promise for treating various diseases such as congenital disorders and viral infections, by correcting the disease-specific genomic sequences.
This technology attracted increasing attention in the last decade as it allows easy targeting of genomic sequences with high precision and low level of undesired off-target gene editing. However, safe and efficient delivery of the Cas9 DNA cutting enzyme and guide RNA (gRNA), which is a short piece of RNA that guides Cas9 to its genomic targets, remains an obstacle.
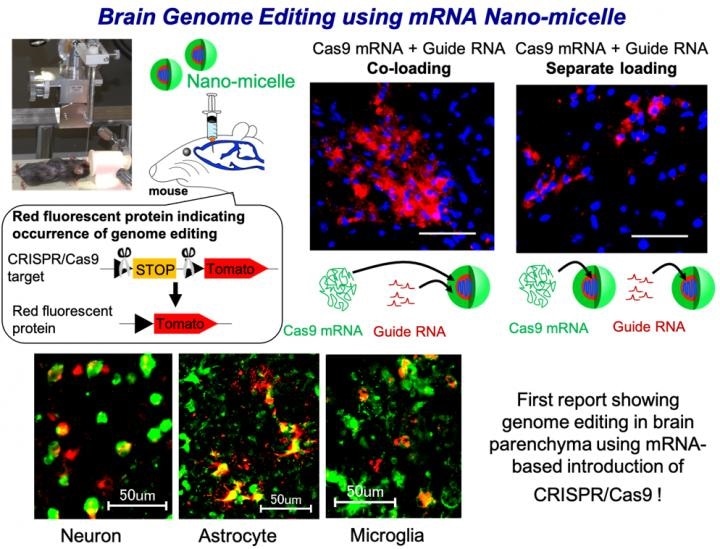
We fabricated nano-micelle for the delivery of Cas9-encoding messenger RNA (mRNA) and gRNA, which condenses these RNAs in the micelle core and forms a protective shell of polyethylene glycol. The efficiency of genome editing was evaluated in transgenic mice which express a red fluorescent protein upon successful genome editing in the mouse brain.
Cas9 mRNA (4500 base) and gRNA (100 base) are largely different in size, and when each of them was loaded separately in a nano-micelle, gRNA was rapidly released and degraded. Interestingly, when both Cas9 mRNA and gRNA were loaded in same nano-micelle, the stability of gRNA was increased and this co-loaded nano-micelle allowed more efficient genome editing in various brain cells, such as neurons, astrocytes and microglia.
To the best of our knowledge, this is the first report validating the utility of RNA-based introduction CRISPR/Cas9 in brain parenchyma. Additionally, the presence of polyethylene glycol on the surface of nano-micelle was found to play a pivotal role in diffusion in the brain tissue after injection, allowing to induce higher genome editing.
Read the original article on News-Medical.


