In a new study published in Science Robotics, led by researchers at the Catalan Institution for Research and Advanced Studies (ICREA), Spain, scientists have observed the collective behaviour of a large number of nanorobots in vivo using radioactive isotope labelling. The nanobots were designed at the Institute for Bioengineering of Catalonia (IBEC), also Spain, to have autonomous movement inside the bladder of living mice.
“Having been able to see how nanorobots move together, like a swarm and of following them within a living organism, is very important, since millions of them are needed to treat specific pathologies such as, for example, cancer tumours,” said Professor Samuel Sánchez, principal investigator at IBEC.
“We have demonstrated for the first time that nanorobots can be monitored in vivo through Positron Emission Tomography (PET), a highly sensitive non-invasive technique used nowadays in the biomedical environment,” explained Jordi Llop, principal investigator at the Radiochemistry & Nuclear Imaging Lab from CIC biomaGUNE, Spain.
For their study, the researchers first carried out in vitro experiments, monitoring the nanorobots through optical microscopy and PET. Both techniques allowed them to observe how the nanoparticles mixed with the fluids and were capable of migrating, collectively, following complex paths. The nanorobots were then administered intravenously to mice and finally introduced into their bladders. Since the nanorobots are coated with an enzyme called urease, which uses the urea from urine as a fuel, they swim collectively inducing fluid flows inside the bladder.
The team found that the distribution of nanodevices in the bladder of the mice was homogeneous, indicating that the collective movement was co-ordinated and efficient.
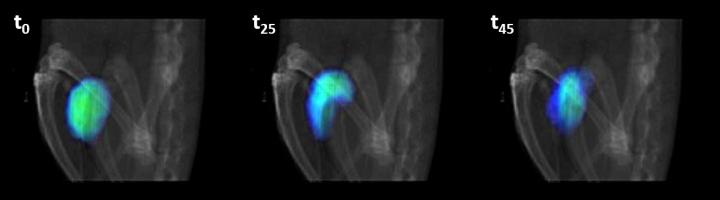
PET-CT imaging obtained 0, 25 and 45 minutes after the nanorobots were delivered in the presence of urea.
“Nanorobots show collective movements similar to those found in nature, such as birds flying in flocks or the orderly patterns that schools of fish follow,” said Sánchez.
“We have seen that nanorobots that have urease on the surface move much faster than those that do not. It is, therefore, a proof-of-concept of the initial theory that nanorobots will be able to better reach a tumour and penetrate it,” said Llop.
“This is the first time that we are able to directly visualise the active diffusion of biocompatible nanorobots within biological fluids in vivo. The possibility to monitor their activity within the body and the fact that they display a more homogeneous distribution could revolutionise the way we understand nanoparticle-based drug delivery and diagnostic approaches,” said Tania Patiño, co-corresponding author of the paper.
Read the original article on Drug Target Review.