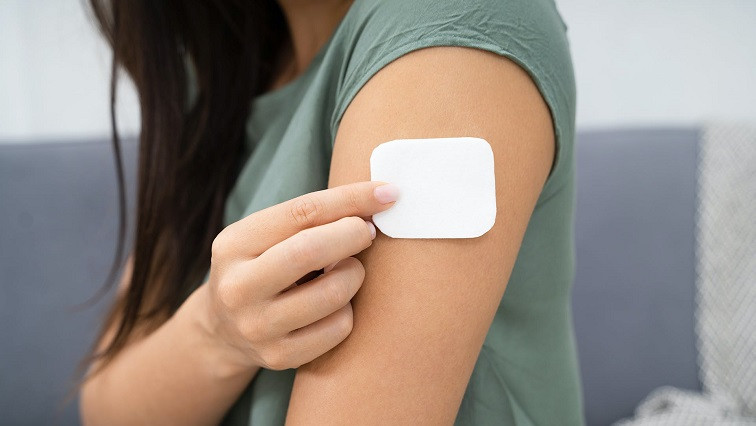
The investment will help expand the company's Hero Patch™ platform, which uses microneedle technology to deliver clinically meaningful doses of macromolecules and small molecules. The deal, which closed on April 8, was led by Josef Volman, who co-chairs the firm’s Business Law Group, and Emmanuel Johnson.
This new class of microneedles was invented at Tufts University and is now the core technology of the Hero Patch™ platform at Anodyne, founded by Hojat Rezaei Nejad, Konstantinos Tzortzakis, and Jake Lombardo. By incorporating solvent-free bulk drug substance directly into microscopic porosities, Anodyne can load unprecedented amounts of the drug onto each microneedle. When the microneedle penetrates into the skin, the drug dissolves in the patient’s interstitial fluid and is systemically absorbed.
“Our set of technologies resolve many challenges that were hindering transdermal delivery of large molecules in a clinically relevant dosage for decades. Anodyne Nanotech’s primary focus is now on biologics, and we have shown delivery of monoclonal antibodies and peptides in vivo. I believe our technology is a gamechanger and unprecedented on many levels, and we continuously work to expand and develop our capabilities,” said Hojat Rezaei Nejad, CTO of Anodyne Nanotech.
“We are proud to be working with Anodyne,” said Josef Volman. “As a Tufts alumnus, it is particularly rewarding to be working with companies that have been launched out of Tufts with such a star team of former students. We look forward to working with Jake and Konstantinos and their impressive investor group on developing a strong business around their cutting-edge science.”
“It has been a very rewarding experience to begin working with Joe in the classroom and continue working with his experienced team at Burns & Levinson. They have played a crucial role in our successful funding rounds that will ultimately propel our development plans,” said Jake Lombardo, CEO of Anodyne.
Not only does Anodyne’s technique allow for unprecedented drug loading, but also using the solid-state form of the drug substance can lead to improved stability. This promise would circumvent the need for cold-chain distribution, a critically important challenge for developing countries, with cost-saving advantages for the global biopharmaceutical industry.
Anodyne has started collaborations with pharmaceutical companies to test particular biologics on the Hero Patch platform. The company plans to conduct clinical trials to characterize the bioequivalence, safety and tolerability of several Hero Patch products.
Read the original article on Burns & Levinson.