As a teenager, I realized I was too sensitive to suffering to become a medical doctor, yet I still wanted to cure the world. I focused on chemistry for my PhD at the University of Miami, Florida, and then pursued physics as a postdoc at Trinity College Dublin, working on carbon nanotubes. The large surface area of nanomaterials gives them unique optical and electrical properties. I learnt that I could add nanoscale ‘switches’ to direct the nanotubes to perform specific functions in human cells. As toxicity concerns grew over nanotubes because of their rigid, hollow shape, I began working on carbon nanoparticles, which have those same desirable properties.
I call these devices nano-onions, because they consist of concentric layers of carbon just 5 nanometres in diameter. I knew that they would be perfect for biomedical applications, because they are small enough to enter a human cell and chemically reactive enough to use in imaging, diagnostics or targeted drug delivery. The one problem was making them soluble in water so that the body could absorb them. My colleague and I did that by adding phospholipids and hyaluronic acid. Cancer cells prefer to bind to hyaluronic acid, so that tricks them into absorbing the nano-onions.
In this photo taken in my laboratory, you can see two key pieces of equipment. In the foreground is the furnace, where we synthesize the nano-onions in a process that I patented. Behind me is the fume cupboard, where we can work safely with the toxic chemotherapeutic compounds that we add to the nano-onions. Although we haven’t commercialized anything yet, I’ve worked for a decade to confirm that these nano-onions have no toxic effects.
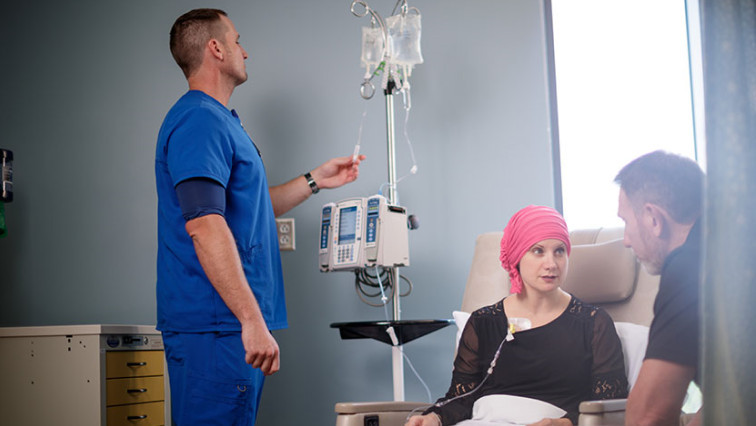
Ideally, my work will improve targeted delivery of chemotherapeutic drugs and avoid adverse effects on healthy cells. I hope to create nanoparticles that can deliver diagnostic, imaging and therapeutic functions — all together.
Read the original article on Nature.