The lithium-ion battery is the future of sustainable energy technology, but drastic volume fluctuations in their anodes related to enhanced battery capacity raises a safety concern. Recently, researchers from the Republic of Korea have found that embedding manganese selenide anodes in a 3D carbon nanosheet matrix is an innovative, simple, and low-cost means of reducing drastic volume expansion while improving the energy density of these batteries.
Lithium-ion batteries (LIBs), which are a renewable source of energy for electrical devices or electric vehicles, have attracted much attention as the next-generation energy solution. However, the anodes of LIBs in use today have multiple inadequacies, ranging from low ionic electronic conductivity and structural changes during the charge/discharge cycle to low specific capacity, which limits the battery’s performance.
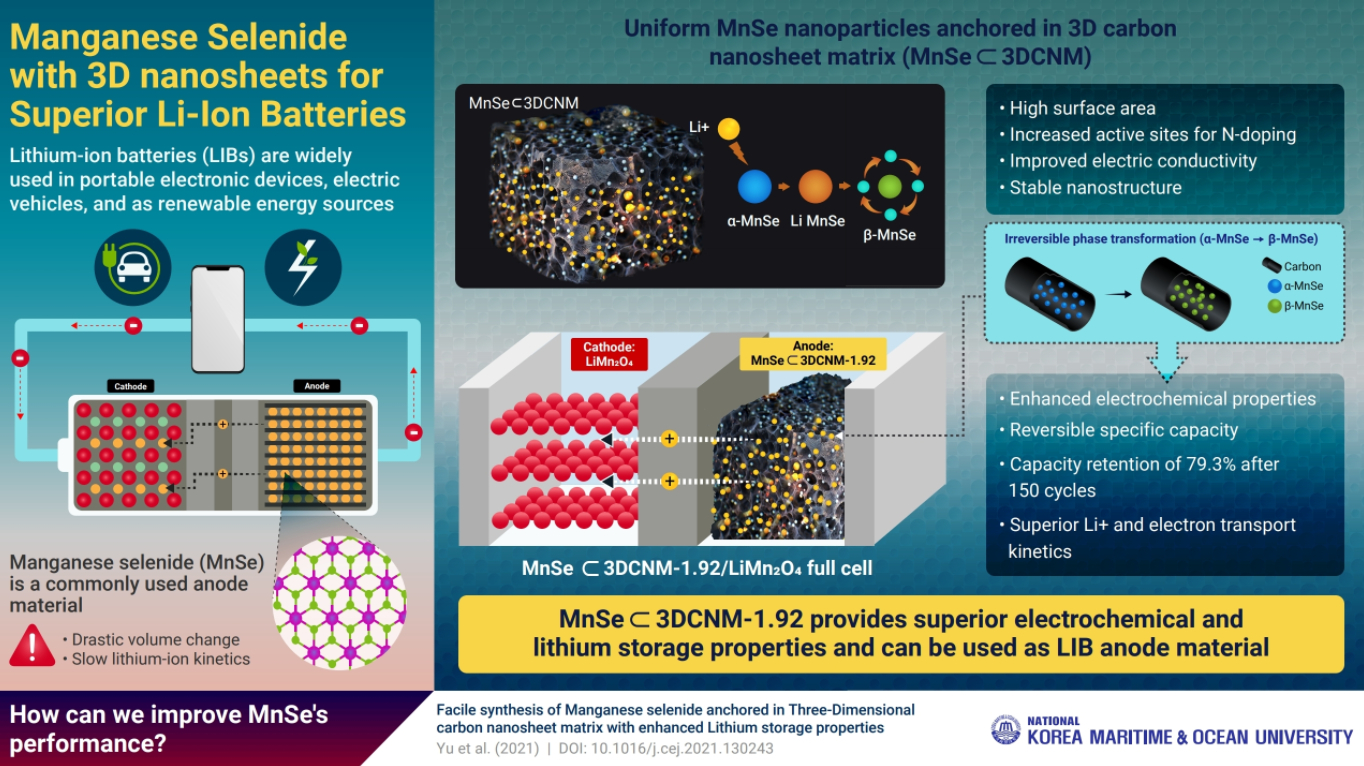
In search of a better anode material, Dr. Jun Kang of Korea Maritime and Ocean University, along with his colleagues from Pusan National University, Republic of Korea, has designed an anode that, owing to its unique structural features, overcomes many of the existing barriers of anodic efficiency. Dr. Kang explains, “We focused on manganese selenide (MnSe), an affordable transition metal compound known for its high electrical conductivity and applicability in developing semiconductors and supercapacitors- as a possible candidate for the advanced LIB anode.” However, MnSe undergoes a drastic volume change (by almost 160%) during the charging-discharging cycles, which not only reduces the performance of the electrode but also raises safety issues.
In an effort to prevent this volume change, the aforementioned researchers developed a simple and low-cost process: they uniformly infused the MnSe nanoparticles into a three-dimensional porous carbon nanosheet matrix (or 3DCNM). In the newly developed anode material (which they termed “MnSe ⊂ 3DCNM”), the carbon nanosheet scaffold endowed the anchored MnSe nanoparticles with numerous advantages, such as a high number of active sites and an enhanced contact area with the electrolyte and protected them from drastic volume expansion.
The researchers were able to synthesize a variety of MnSe ⊂ 3DCNM materials. Among these, they found MnSe ⊂ 3DCNM-1.92 to exhibit the best cycle stability and rate capabilities. When combined with lithium manganese (III,IV) oxide (LiMn2O4, a commonly used cathode material) in a full cell, the team observed that MnSe ⊂ 3DCNM-1.92 remarkably continued to demonstrate superior electrochemical properties, including superior lithium ion and electron transport kinetics!
The team is excited about the potential implications of their accomplishment. As Dr. Kang explains, “Using a conducive filler scaffold, we have developed an anode that boosts the battery performance while simultaneously allowing reversible energy storage. This strategy can serve as a guide for other transition metal selenides with high surface areas and stable nanostructures, with applications in storage systems, electrocatalysis, and semiconductors.”
Along with this new development in the field of LIBs, the possibility of realizing a greener future becomes brighter!
Read the original article on Korea Maritime and Ocean University.







