“Through our research, we have succeeded in binding water-insoluble antibacterial molecules to the graphene and having the molecules released in a controlled, continuous manner from the material. This is an essential requirement for the method to work. The way in which we bind the active molecules to the graphene is also very simple, and could be easily integrated into industrial processes,” explains Santosh Pandit, a researcher at the Department of Biology and Biological Engineering at Chalmers, and first author of the study which was recently published in Scientific Reports.
Certain bacteria can form impenetrable surface layers, or ‘biofilms’, on surgical implants, such as dental and other orthopaedic implants, and represent a major problem for healthcare globally. Biofilms are more resistant than other bacteria, and the infections are therefore often difficult to treat, leading to great suffering for patients, and in the worst cases, necessitating removal or replacement of the implants. In addition to the effects on patients, this entails large costs for healthcare providers.
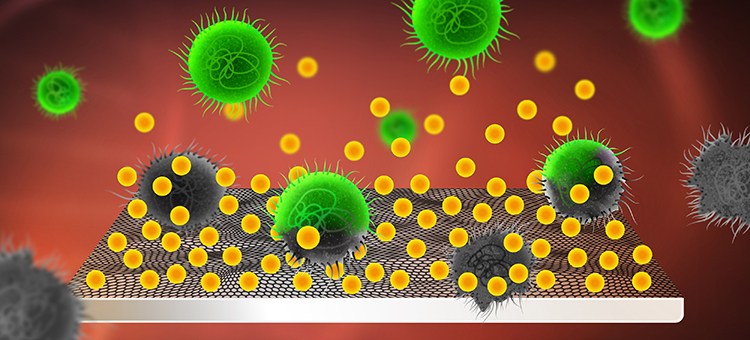
Usnic acid (yellow) is integrated in and released from the implant’s graphene coating. The usnic acid kills the bacteria (green) and thereby prevents them from forming infectious biofilms on the surface.
Graphene is suitable as an attachment material
There are a variety of water-insoluble, or hydrophobic, drugs and molecules that can be used for their antibacterial properties, but in order for them to be used in the body, they must be attached to a material, which can be difficult and labour intensive to manufacture.
“Graphene offers great potential here for interaction with hydrophobic molecules or drugs, and when we created our new material, we made use of these properties. The process of binding the antibacterial molecules takes place with the help of ultrasound,” says Santosh Pandit.
In the study, the graphene material was covered with usnic acid, which is extracted from lichens, for example, fruticose lichen. Previous research has shown that usnic acid has good bactericidal properties. It works by preventing bacteria from forming nucleic acids, especially inhibiting of RNA synthesis, and thus blocking protein production in the cell. Usnic acid was tested for its resistance to the pathogenic bacteria Staphylococcus aureus and Staphylococcus epidermidis, two common culprits for biofilm formation on medical implants.
Simple method paves way for future drugs
The researchers’ new material displayed a number of promising properties. In addition to successful results for integrating the usnic acid into the surface of the graphene material, they also observed that the usnic acid molecules were released in a controlled and continuous manner, thus preventing the formation of biofilms on the surface.
“Even more importantly, our results show that the method for binding the hydrophobic molecules to graphene is simple. It paves the way for more effective antibacterial protection of biomedical products in the future. We are now planning trials where we will explore binding other hydrophobic molecules and drugs with even greater potential to treat or prevent various clinical infections,” says Santosh Pandit.
Read the original article on Chalmers University of Technology.







