Precise metal nanoclusters (NCs) are ideal for developing practical catalysts for chemical reactions. However, their catalytic activity is reduced either due to protective molecules called "ligands" surrounding them or aggregation resulting from ligand removal. In a new study, scientists from Japan elucidate the ligand removal mechanism for gold NCs and irradiate them with UV light to prevent aggregation, creating a high-functioning photocatalyst.
Catalysts, which help drive reactions, are ubiquitous, whether as an enzyme in the body that digests food or the catalytic converter in the car that breaks down pollutants. In chemical reactions, catalysts play an important role in making it more efficient. Recently, atomically precise metal nanoclusters (NCs) that can accelerate various thermal, electrochemical, and photochemical reactions have been used to design useful catalysts. These NCs are tiny particles (less than 2 nanometers) whose properties can be modified by changing their atomic composition. This is why, metal NCs have received considerable attention, with scientists trying to find various ways of synthesizing NCs with unique functions.
A popular way of fabricating atomically precise metal NCs is using ligands (molecules or ions that attach themselves to a central metal core). These ligands not only protect the tiny NCs but also affect their chemical reactivity and selectivity. Sometimes, however, the reactivity is lower than expected.
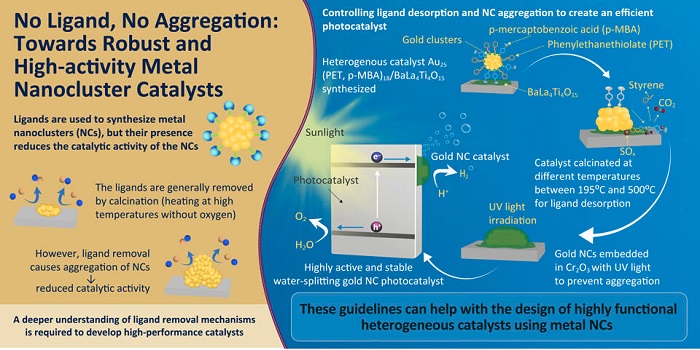
To increase the catalytic activity of ligand-protected metal NCs, they are heated in a furnace at high temperatures without oxygen (a process called "calcination") to remove the ligands from the main cluster. However, heating the particles at very high temperatures can cause the NCs to accumulate, often leading to a decrease in reactivity. "When the ligands are removed without special treatment, the metal NCs easily aggregate on the support and lose their size-specific properties. It is essential understand the mechanism of ligand calcination to create highly functional heterogeneous catalysts under appropriate conditions," says Prof. Yuichi Negishi of Tokyo University of Science, Japan, who researches on the synthesis of nanoclusters.
In a new study published in Angewandte Chemie, Prof. Negishi led a team of researchers, including Assistant Professor Tokuhisa Kawawaki, Mr. Yuki Kataoka, Ms. Momoko Hirata, and Mr. Yuki Akinaga, to dig deep into the mechanism of the ligand removal process in NCs. For their experiments, the researchers synthesized gold NCs protected by two ligands, 2-phenylethanethiolate and mercaptobenzoic acid and then supported them on a photocatalytic metal oxide. Next, the team heated the prepared material at different temperatures ranging from 195°C to 500°C. After every step, they analyzed the products using techniques such as infrared spectroscopy, x-ray photoelectron spectroscopy, and transmission electron microscopy to identify the changes in their chemical composition.
After the ligands were completely released, the team embedded the gold NCs within a thin film of chromium oxide by irradiating the sample with UV light in order to prevent aggregation of the NCs. This process generated a photocatalyst with useful properties like high water-splitting activity and stability.
These findings guide the design for metal NC-based catalysts in the future, with applications in hydrogen generation for hydrogen fuel cells. "With our research, we hope to build a clean, sustainable, society, one brick at a time," concludes Prof. Negishi.
Read the original article on Tokyo University of Science.






