Introduction
Nucleic acid therapeutics can harness the potential of genetics to treat human diseases. Because nucleic acid drugs can be targeted on diverse proteins, there are infinite possible therapeutic applications in this new paradigm of treatment.
Nucleic acids such as deoxyribonucleic acid (DNA) or mRNA can be delivered to replace mutated genes in rare genetic diseases. Comparatively, short interfering RNAs (siRNAs) and antisense oligonucleotides (ASOs) can be tailored to knock down harmful mutant proteins in cancers. While conventional therapies are unsuccessful in most of these cases, nucleic acid therapeutics show enormous promise.
As evidenced in the recent efforts to mitigate the ongoing coronavirus disease 2019 (COVID-19) pandemic, mRNA technology has been used in several novel vaccines to induce an immune response comprising both cellular and humoral immunity.
However, challenges such as efficient delivery, stability, and safe use of naked nucleic acids have hindered its successful clinical applications. The current development of nonviral LNPs, which have already been administered to hundreds of millions of individuals around the world in the form of the COVID-19 mRNA vaccines, helps to overcome the innate barriers to nucleic acid delivery.
Therapeutic nucleic acids
Underlying cellular dysfunction can be addressed using nucleic acids such as RNA, which is an intermediate between the genome and the protein. Unlike DNA, which needs to enter the nucleus to transcribe, RNA acts in the cytosol to translate into a protein.
Currently, the most clinically advanced therapies using nonviral delivery vectors include therapeutic siRNA or mRNA. When a toxic or harmful protein due to a mutation needs to be knocked down to treat a disease, the siRNA tool is used. The siRNA approach was discovered in 1998 in Caenorhabditis elegans, which opened up a new molecular technique to knock down a specific protein.
Similarly, when a gene with a null mutation needs to be upregulated or replaced to fix the expression of a vital protein, mRNA is used. With essential modifications, the desired mRNA can be synthesized using commercial enzymes through the process of in vitro transcription (IVT), which is also readily scalable.
Delivery barriers and nucleic acid modifications
Despite the recognition of the therapeutic use of nucleic acids, several barriers have impeded its potential use.
RNA is vulnerable to degradation by the immune system, as the naked RNA activates pattern-recognition receptors (PRR) as a pathogen-associated molecular pattern (PAMP). When the single-stranded RNA (ssRNA) and double-stranded RNA (dsRNA) activate Toll-like Receptors (TLR) as part of the innate immune system, it leads to inflammation and heightened immunogenicity.
These barriers are overcome by modifications to the RNA, such as to incorporate noncanonical and chemically modified nucleoside bases outside of the Watson–Crick bases (A, G, C, U). Replacing the uracil with pseudouridine (Ψ) in an mRNA sequence results in reduced immune activation, in addition to an improved translation of the desired protein. Additionally, modifications with 1-methylpseudouridine (1mΨ) and 5-methoxyuridine (5moU) improve sequence translation and overall stability, though presented cell-type differences in protein expression.
Importantly, these therapeutic approaches require the use of high purification methods to remove any trace of impurities to reduce undesired immune reactions and improve overall delivery. “Although the reactogenicity caused by nucleic acids can be beneficial in activating the immune response for certain vaccine technologies,” noted the reviewers.
RNA can also be degraded by RNases and endonucleases within the serum when delivered systemically or intravenously (IV). The RNA or the nanoparticle delivery vehicles must also bypass the reticuloendothelial system (RES) and the liver, which selectively uptakes particles in the 80–200 nanometer (nm) size range.
Because nucleic acids are large molecules and are strongly negatively charged due to the sugar-phosphate backbone, a robust delivery strategy is required. Thus, delivery vehicles must not only protect RNA from immune response and degradation but also facilitate intracellular uptake.
Nonviral delivery vectors and lipid nanoparticles
While viruses act as potent vectors for cellular transfection, recent efforts have expanded the use of nonviral nanoparticle vectors.
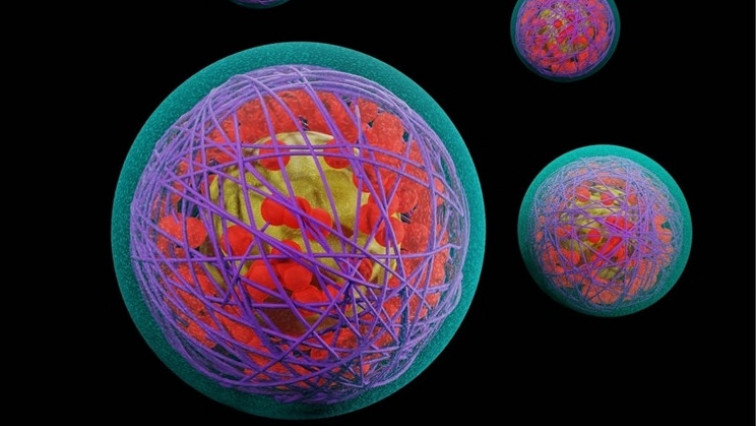
Using microfluidic mixing, scalable and replicable laboratory- or industrial-scale LNPs are produced. LNPs are a combination of four chemical components including phospholipids, cholesterol, lipid anchored polymers, and an ionizable lipid.
The phospholipids and cholesterol form a lipid bilayer to encapsulate the nucleic acid cargo. Comparatively, the lipid-anchored polymers typically contain hydrocarbon lipid tails and long poly(ethylene glycol) (PEG) polymeric chains that prevent the non-specific uptake of nanoparticles. The ionizable lipid component is essential for the complexation and encapsulation of the RNA cargo and also assists in endosomal escape and intracellular delivery.
The LNPs and their synergistic formulations have been used in many drug delivery systems to treat diseases. Only about 2–10% of LNP release their cargo to the cytosol. However, a substantial impact on the protein levels can be achieved, as the delivery of even a few siRNAs or mRNAs can be repeatedly used in the inhibition or initiation of translation respectively.
Design of mRNA vaccines to combat COVID-19
Nucleic acid vaccines deliver the genetic sequence required to express a viral protein or antigen within the body, thus triggering an immune response. Notably, before 2020, there were no clinical trials for mRNA vaccines, despite successful preclinical results.
Utilizing an LNP delivery strategy, within one year of sequencing the SARS-CoV-2 genome, strongly efficacious mRNA vaccines were developed. These are also currently the only mRNA therapeutics currently being used in patients. Reports highlight that these vaccines protect against severe disease outcomes at rates of about 97% or higher in terms of their ability to reduce hospitalizations and deaths.
Outlook and future potential
The development of LNPs as delivery vehicles for nucleic acid therapeutics launches a new paradigm to rapidly respond to global health problems.
Novel mRNA transcripts can be generated to respond to emerging variants or future pathogens. The same LNP technology can be expedited to combat SARS-CoV-2 variants without the need for clinical trials. LNPs can also be used for other threats, including cancer, through vaccine-mediated immunotherapy.
Additional improvements in ionizable lipid design, RNA technology (such as self-amplifying mRNA which requires less material), and industrial manufacturing may soon lead to widespread benefits of RNA delivery, the reviewers write.
Read the original article on News Medical.