Living organisms, viruses, and technological devices, have water layers between their cells or parts and can die or stop working when dehydrated. But why water and not any other fluid? What makes water unique under such conditions when it is in these tiny structures?
“It all depends on the peculiar hydrogen bond interaction of water and this might be a key factor contributing to the solution of one of the Sustainable Development Goals defined by the United Nations, the one about clean water and sanitation”, explains Giancarlo Franzese, from the Institute of Nanoscience and Nanotechnology of University of Barcelona (IN2UB).
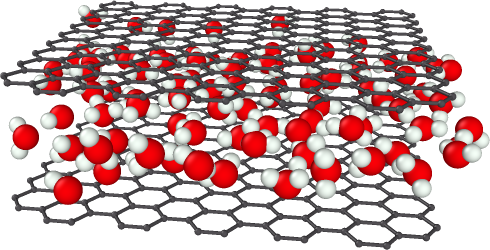
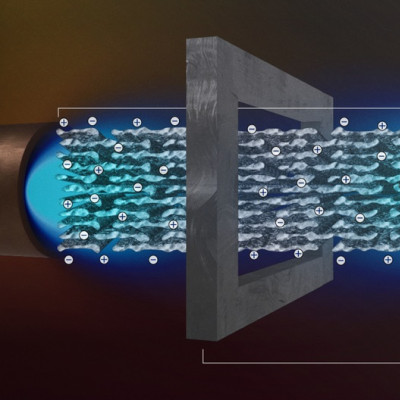
Actually, in water treatment and sanitation, there is a lot of research on the nanometer-size graphene pores. Furthermore, the result of this joint project between the UB and La Sapienza relates to the switching behaviour observed in a hydrated graphene nano-memristor, that is a nanotechnology device where the electric charge rules the magnetic flux, when the confined water changes from one to more layers.
“Nanoconfinement can drastically change the behaviour of liquids, puzzling us with counterintuitive properties. It is relevant in applications, including decontamination and crystallization control”, says Carles Calero from the IN2UB. In the study, researchers compare, using molecular dynamics simulations, three different liquids in a graphene slit pore: a simple fluid, such as argon; a molecular fluid, such as CO2 or a liquid metal, and water. The three fluids, under similar subnanometric confinement, behave differently from each other, opening the way for possible applications with nanopores, for example, for the elimination of pollutants.
Read the original article on University of Barcelona.