Bioelectricity, the current that flows between our cells, is fundamental to our ability to think and talk and walk. In addition, there is a growing body of evidence that recording and altering the bioelectric fields of cells and tissue plays a vital role in wound healing and even potentially fighting diseases like cancer and heart disease.
Now, for the first time, researchers at the USC Viterbi School of Engineering have created a molecular device that can do both: record and manipulate its surrounding bioelectric field.
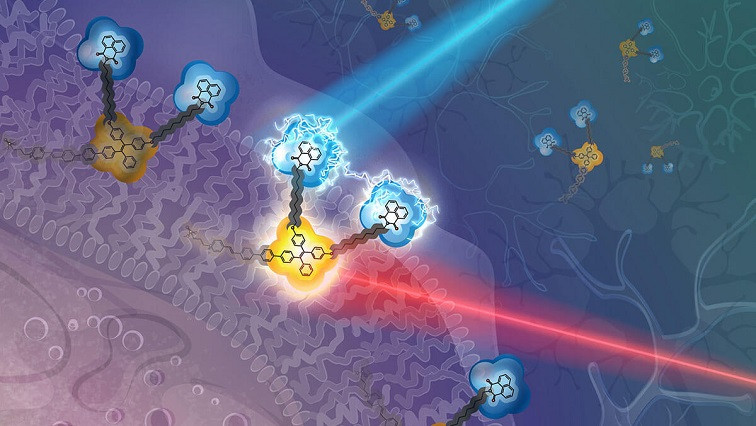
The triangle-shaped device is made of two small, connected molecules — much smaller than a virus and similar to the diameter of a DNA strand.
It’s a completely new material for “reading and writing” the electric field without damaging nearby cells and tissue. Each of the two molecules, linked by a short chain of carbon atoms, has its own separate function: one molecule acts as a “sensor” or detector that measures the local electric field when triggered by red light; a second molecule, “the modifier,” generates additional electrons when exposed to blue light. Notably, each function is independently controlled by different wavelengths of light.
Though not intended for use in humans, the organic device would sit partially inside and outside the cell’s membrane for in vitro experiments.
The work, published in the Journal of Materials Chemistry C, was spearheaded by USC Viterbi professors Andrea Armani and Rehan Kapadia. The lead authors include Yingmu Zhang, a postdoctoral researcher in the Mork Department of Chemical Engineering and Material Science; and Jinghan He, a Ph.D. candidate in the USC Department of Chemistry. Co-authors include Patrick Saris, USC Viterbi postdoctoral researcher; and Hyun Uk Chae and Subrata Das, Ph.D. candidates in the Ming Hsieh Department of Electrical and Computer Engineering. The Armani Lab was responsible for creating the new organic molecule, while the Kapadia Lab played a key role in testing how efficiently the “modifier” was generating electricity when activated by light.
Because the reporter molecule can insert into tissue, it has the possibility to measure electric fields non-invasively, providing ultra-fast, 3-D, high resolution imaging of neural networks. This can play a crucial role for other researchers testing the effects of new drugs, or changes in conditions like pressure and oxygen. Unlike many other previous tools, it will do so without damaging healthy cells or tissue or requiring genetic manipulation of the system.
“This multi-functional imaging agent is already compatible with existing microscopes,” said Armani, the Ray Irani Chair in Chemical Engineering and Materials Science, “so it will enable a wide range of researchers — from biology to neuroscience to physiology — to ask new types of questions about biological systems and their response to different stimuli: drugs and environmental factors. The new frontiers are endless.”
In addition, the modifier molecule, by altering the nearby electric field of cells, can precisely damage a single point, allowing future researchers to determine the cascading effects throughout, say, an entire network of brain cells or heart cells.
“If you have a wireless network in your home, what happens if one of those nodes becomes unstable?” said Armani. “How does that affect all the other nodes in your house? Do they still work? Once we understand a biological system like the human body, we can better predict its response – or alter its response, such as making better drugs to prevent undesirable behaviors.”
“The key thing,” said Kapadia, the Colleen and Roberto Padovani Early Career Chair in Electrical and Computer Engineering, “is that we can use this to both interrogate as well as manipulate. And we can do both things at very high resolutions – both spatially and temporally.”
Key to the new organic device was the ability to eliminate “crosstalk.” How to get these two very different molecules to join together and not interfere with each other in the manner of two scrambled radio signals? In the beginning, notes Armani, “it wasn’t entirely obvious that it was even going to be possible.” The solution? Separate both by a long alkyl chain, which does not affect the photophysical abilities of each.
Next steps for this multi-functional new molecule include testing on neurons and even bacteria. USC scientist Moh El-Naggar, a collaborator, has previously demonstrated the ability of microbial communities to transfer electrons between cells and across relatively long distances — with huge implications for harvesting biofuels.
Read the original article on University of Southern California.