This approach demonstrates a general strategy for synthesizing MPEI nanoparticles, not only providing a step toward octonary intermetallics, but also enabling the synthesis of MPEIs at the nanoscale.
Nano-MPEIs are intermetallic compounds – roughly 4-5 nanometers diameter – composed of multi-elemental metals in definite proportions, as opposed to alloy, for example, which has variable proportions. The properties and crystal structure formed by these intermetallic compounds are different from its constituents.
The study, led by Liangbing Hu, Herbert Rabin Distinguished Professor of Materials Science and Engineering (MSE) at the University of Maryland (UMD) and director of the Center for Materials Innovation (CMI) – was published in Science Advances on January 28, 2022. Mingjin Cui, a former MSE Faculty Assistant at UMD, served as the first author on the research paper.
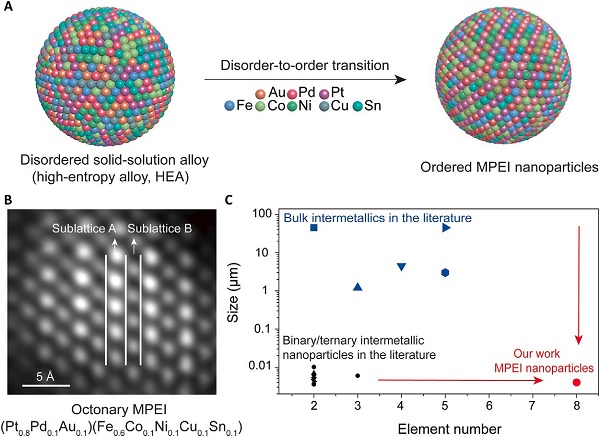
MPEI nanoparticle synthetic process, product and comparison with the literature.
“Precisely controlling the atomic ordering arrangement inside nanoparticles is a great challenge,” said Hu. “We achieved the multi-elemental ordered intermetallic nanoparticles by unique disorder-to-order transition strategy. The strategy can be extended to produce a combinatorial library of intermetallic nanomaterials that may feature novel properties and applications.”
To achieve this, the group rapidly heated metal-salt precursors on a carbon substrate at 1100 K. Once cooled, they reheated the nanoparticles for five minutes, again at 1100 K, to encourage atomic rearrangement (thus ensuring the elements would fall in proper order), which gave way to a more stable MPEI arrangement. Then, the elements were rapidly cooled. The result was MPEI nanoparticles boasting an intermetallic structure with multiple elements.
“What makes this work special is the nano-MPEIs in this case demonstrate high activity and stability in catalysis,” said Cui. “Nano-MPEIs have not been achieved previously by traditional wet chemistry or long-time sintering processes.”
Not only can this process bolster applications in catalysis, magnetics and superconductors, but the group expects to create a library, so to speak, of nanoparticle MPEIs, which could potentially give way to a new frontier of intermetallic research and applications.
Read the original article on University of Maryland (UMD).