Scientists in New Zealand and Australia working at the level of atoms created something unexpected: tiny metallic snowflakes.
Why’s that significant? Because coaxing individual atoms to cooperate is leading to a revolution in engineering and technology via nanomaterials. (And creating snowflakes is cool.)
Nanoscale structures (a nanometre is one billionth of a metre) can aid electronic manufacturing, make materials stronger yet lighter, or aid environmental clean-ups by binding to toxins.
To create metallic nanocrystals, New Zealand and Australian scientists have been experimenting with gallium, a soft, silvery metal which is used in semiconductors and, unusually, liquifies at just above room temperature.
Their results were just reported in the journal Science.

This metallic snowflake is about 100 microns across, roughly the thickness of a human hair.
Professor Nicola Gaston and research fellow Dr Steph Lambie, both of Waipapa Taumata Rau, University of Auckland, and Dr Krista Steenbergen of Te Herenga Waka, Victoria University of Wellington, collaborated with colleagues in Australia led by Professor Kourosh Kalantar-Zadeh at the University of New South Wales.
The Australian team worked in the lab with nickel, copper, zinc, tin, platinum, bismuth, silver and aluminium. Metals were dissolved in gallium at high temperatures. Once cooled, the metallic crystals emerged while the gallium remained liquid.
The New Zealand team, part of the MacDiarmid Institute for Advanced Materials and Nanotechnology, a national Centre of Research Excellence, carried out simulations of molecular dynamics to explain why differently shaped crystals emerge from different metals. (The government’s Marsden Fund supported the research.)
“What we are learning is that the structure of the liquid gallium is very important,” says Gaston. “That’s novel because we usually think of liquids as lacking structure or being only randomly structured.”
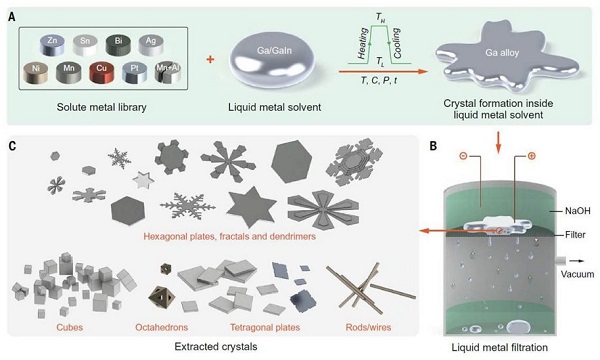
Interactions between the atomistic structures of the different metals and the liquid gallium cause differently shaped crystals to emerge, the scientists showed.
The crystals included cubes, rods, hexagonal plates and the zinc snowflake shapes. The six-branched symmetry of zinc, with each atom surrounded by six neighbours at equivalent distances, accounts for the snowflake design.
“In contrast to top-down approaches to forming nanostructure – by cutting away material – this bottom-up approaches relies on atoms self-assembling,” says Gaston. “This is how nature makes nanoparticles, and is both less wasteful and much more precise than top-down methods.”
She says the research has opened up a new, unexplored pathway for metallic nanostructures. “There’s also something very cool in creating a metallic snowflake!”
Read the original article on University of Auckland.







