Phototherapy techniques such as photodynamic therapy (PDT) and photothermal therapy (PTT) have generated extensive interest as promising therapeutic techniques for the treatment of cancer because of their minimal invasiveness. In PDT, photosensitizers are activated to produce reactive oxygen species to destroy cancer cells upon light irradiation. For PTT, a photothermal agent is used to absorb and convert light radiation energy into heat to destroy cancer cells. However, conventional PDT suffers from the “always-on” mode with severe post-treatment side effects and inefficient generation of reactive oxygen species. PTT treatment also has several disadvantages such as unnecessary damage caused by overheating and relatively low efficiency in the delivery of photothermal agents to the targeted site.
A research team led by Prof XU Qing-Hua from the Department of Chemistry, National University of Singapore designed an aggregation-induced emission enhancement scheme for the photosensitizer, which is based on conjugated metal nanoparticles. These nanoparticles display multimodal imaging capabilities and have highly efficient PDT/PTT synergetic therapy effects in their aggregated form. In this approach, aggregates of Chlorin e6 conjugated gold nanoparticles were demonstrated to display significantly enhanced one- and two-photon excitation fluorescence, singlet oxygen generation and photothermal effects. The photoactivity of the photosensitizers was first suppressed by attaching them to gold nanoparticles but it became significantly enhanced when they formed aggregates. In addition to increased brightness, the photostability of the nanoparticle aggregate improved significantly when compared to their non-aggregated form. This is due to the shortened lifetime of the molecules in their excited state.
Based on their findings, the researchers have demonstrated multimodal imaging and synergistic phototherapy using HeLa cancer cells under laboratory conditions. By forming aggregates in-situ inside cancer cells, the nanoparticle aggregates act as fluorescence imaging probes under one- and two-photon excitation using red and near-infrared light, respectively. These nanoparticle aggregates also displayed excellent PDT and PTT performance which can effectively destroy cancer cells by using red light lasers with power density that is below the skin tolerance threshold owing to the synergistic therapy effects.
Prof Xu said, “This plasmonic nanosystem, which is based on a new type of aggregation-induced emission phenomenon, offers a promising technique for imaging guided PDT/PTT synergetic therapy. This could potentially open up a new venue to explore safe, accurate and noninvasive theranostics.”
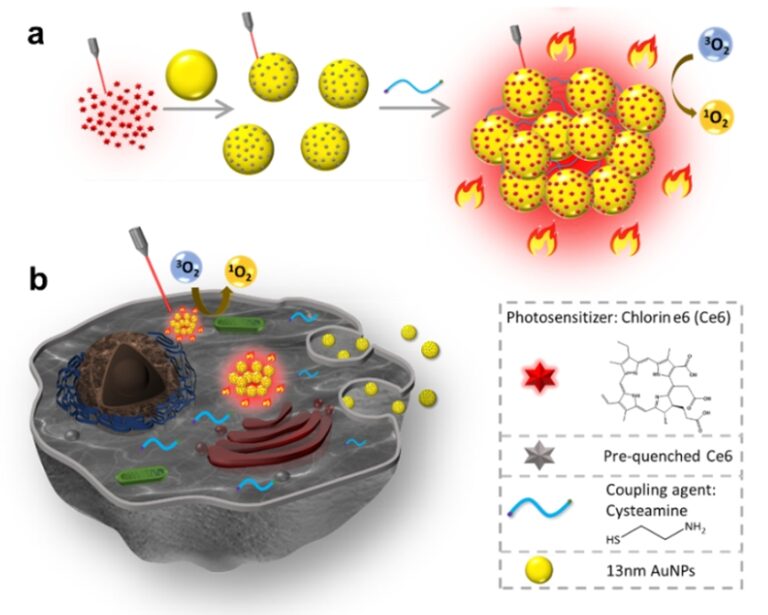
Figure describes the imaging and phototherapy capability of aggregated Ce6 conjugated gold nanoparticles in (a) solution and in (b) cells. Strong red emission under near-infrared irradiation allows cancer cells to be easily imaged. Synergistic photodynamic and photothermal therapy effects (illustrated by the flame pattern) enables the effective killing of cancer cells using red light with power density below the skin tolerance threshold.
Read the original article on National University of Singapore.







