Due to exactly atomic composition and crystal structure, atomically precise copper nanoclusters are used as model catalysts for catalytic reaction mechanisms study. However, the efficient synthesis of copper nanoclusters still remains challenging.
In a recent study published in Chemical Science, a research group led by Prof. HUANG Jiahui and Prof. FAN Hongjun from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) has developed a new strategy for the synthesis of atomically precise copper nanoclusters by solvent-mediated precipitation.
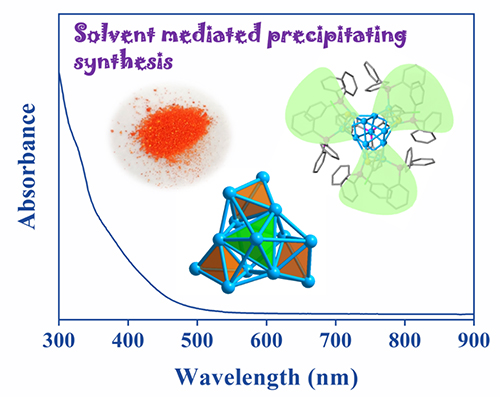
The total crystal structure and absorption optical spectrum of Cu13H10(SR)3(PPh3)7 nanoclusters.
"We named the new strategy as 'solvent-mediated precipitation synthesis (SMPS)', it was realized to efficiently synthesize Cu13H10(SR)3(PPh3)7 nanoclusters where SR was a thiolate ligand," said Prof. HUANG.
The copper nanoclusters, Cu13 for short, were prepared via precipitation, and the excess raw materials and by-products were remained in the solution, solving the problems of separation and low yield of Cu nanoclusters.
Single crystal X-ray diffraction analysis showed that the core of the Cu13 nanocluster with a triple axis of symmetry consists of four vertices-sharing tetrahedrons. Electrospray ionization mass spectrometry and nuclear magnetic resonance spectroscopy confirmed the types and number of hydrogen atoms in Cu13 nanoclusters. Density functional theory calculations simulated the location of 10 hydrides in the crystal structure.
Moreover, steady-state ultraviolet-visible absorption and fluorescence spectra of Cu13 nanoclusters exhibited unique optical absorbance and photoluminescence.
Read the original article on Chinese Academy of Sciences (CAS).