Vascular drug therapies often lead to undesirable side effects on healthy tissues. Addressing this issue requires targeted drug delivery systems. Scientists from Japan have recently developed an innovative approach for measuring the amount of molecules desorbed from the surface of individual microbubbles when irradiated with ultrasound. Using this technique, they analyzed the desorption process in detail, revealing insights that could enable the design of next-generation drug delivery systems based on drug-loaded microbubbles and ultrasound.
Injecting drugs into the bloodstream can often harm healthy tissues as well. Drug delivery systems (DDSs) are an innovative solution designed to target specific cells and minimize such side effects. One strategy for drug delivery that has steadily gained traction involves a combination of microbubbles and ultrasound. Microbubbles are small gas-filled bubbles that can be loaded with drugs or other therapeutic agents on their surface. When exposed to ultrasound waves, these microbubbles begin to oscillate, with the ensuing vibrations gradually releasing the adsorbed drug at the irradiated location. Although this phenomenon has been studied in the past, quantitative experiments on how molecules desorb from microbubbles upon ultrasound irradiation are scarce.
In a recent study published on 22 August 2023 in the journal Scientific Reports, a research team headed by Professor Daisuke Koyama and graduate student Ms. Reina Kobayashi from the Faculty of Science and Engineering at Doshisha University, Japan, sought to address this knowledge gap. They designed an innovative experimental setup to analyze the desorption of a surfactant from individual microbubbles. Their work also included contributions from Dr. Marie Pierre Krafft from Institut Charles Sadron (CNRS), University of Strasbourg, France.
“In a DDS using ultrasound and microbubbles, microbubbles having drugs or genes are injected into blood vessels so that the bubbles can adsorb to the target tissue specifically via antigen-antibody reactions. Our proposed method and the experimental results can predict how much molecules are desorbed from the bubbles into blood vessels,” explains Prof. Koyama.
The method proposed is based on measuring the contact angle of individual bubbles placed on a glass plate using a high-speed video camera. The contact angle, or the angle formed between the solid and bubble surfaces at their point of contact, is closely related to the bubble’s surface tension. In turn, surface tension depends on the amount of surfactant, i.e., drug molecules, on the bubble’s surface. Thus, by observing the contact angle of a bubble under ultrasound irradiation, it is possible to estimate the amount of molecules desorbed as a result of the induced vibrations.
To test their methodology, the team devised an experimental setup combining a high-speed camera with a long-distance microscope, an ultrasound cell filled with fluid, a thin, transparent glass plate, and a laser Doppler vibrometer (LDV) paired with a CCD camera. They carefully placed microbubbles composed of fluorocarbon-enriched gas and a lipid called 1,2-dimyristoyl-sn-glycero-3-phosphocholine (DMPC) onto the glass plate. While the high-speed camera provided real-time data on the bubble’s contact angle, the LDV and CCD camera allowed the researches to monitor the amplitude of the oscillations induced on the bubble as well as its radius.
Using this setup, the researchers measured the desorption characteristics for different bubble sizes and DMPC concentrations, revealing important insights on the process of ultrasound-induced desorption. In particular, they discovered that microbubbles under resonant conditions release significant amounts of adsorbed molecules (>50%) into the surrounding media very quickly. They also demonstrated that ultrasound-induced desorption is a very rapid process, starting almost immediately upon ultrasound irradiation and ceasing just as fast. In addition, they showed that the amount of molecular desorption induced by ultrasonication depends on the bubble size, meaning that the vibrational characteristics of microbubbles used in DDSs are important for controlling drug release.
The proposed method could prove essential in the design and development of DDSs using ultrasound and microbubbles. “The amounts of drug released into blood vessels in patients can be estimated quantitatively using our method, meaning that the optimal amounts of drug-carrying microbubbles can be accurately predicted for vascular drug therapies,” clarifies Prof. Koyama. By administering only the required amount of drug into the bloodstream, the side effects can be kept at a minimum, improving patient outcomes and quality of life.
The research team plans to further explore controlled drug release by clarifying the relationship between the ultrasound frequency, sound pressure amplitude, and the quantity of molecular desorption in their future work. Let us hope their efforts will pave the way to more effective therapeutic options with lesser side effects!
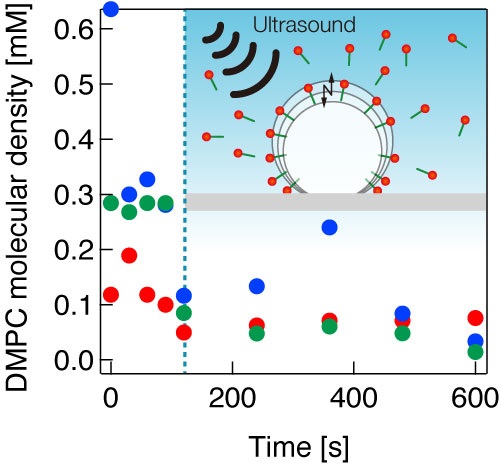
Molecular desorption from a microbubble under ultrasound irradiation.
The amount of molecular desorption can be estimated quantitatively by the proposed method.
Read the original article on Doshisha University.







