Since the discovery of carbon nanotubes (CNTs) 30 years ago, already tons of these are produced each year and find application in different fields, for example in batteries, filter systems, or bicycle frames. Usually, the synthesis of carbon nanotubes takes place at temperatures above 700 °C using pure metal catalysts. But, how to make the synthesis more sustainable and open the way for new application possibilities?
Prof. Joohyun Lim and Prof. Christina Scheu from the independent research group “Nanoanalytics and Interfaces” at the Max-Planck-Institut für Eisenforschung (MPIE) and their colleagues from the Yonsei University in Seoul (Korea) published their recent findings in the journal Advanced Functional Materials.
“We found that the catalytic properties for the synthesis of carbon nanotubes depend on the structure of the catalyst and, therefore, a detailed analysis using electron microscopy is needed to optimize the material”, explains Prof. Lim who very recently returned to South Korea where he started as an assistant professor at the Department of Chemistry at the Kangwon National University.
In their study, the researchers used MnO2 catalyst materials with different amounts of Fe ions implemented, which enabled a remarkably higher catalytic activity for CNT growth at a temperature of only 400-500 °C. “Preparing CNTs at lower temperatures would allow for a broadening of possible applications, for example in solar cells.”
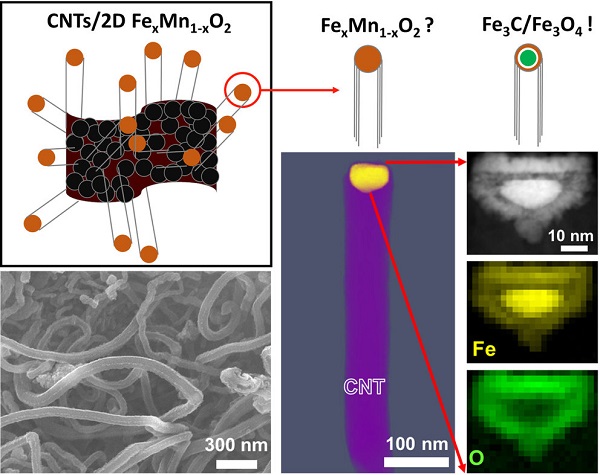
Electron micrograph of carbon nanotubes (lower left), chemical mapping of catalyst particles (lower right) and refined schematic of carbon nanotube growth (top).
By a combination of several techniques and especially (scanning) transmission electron microscopy, the team around Joohyun Lim has been able to investigate the catalyst material in detail. Using energy-dispersive X-ray spectroscopy and electron energy loss spectroscopy they found out how the elemental composition of the catalyst for example influences the overall performance and also the length of the grown CNTs.
The scientists at the MPIE were able to investigate catalyst particles in three dimensions to even better understand their catalytic behaviour. Particles still attached to the CNTs, which grew from them, allowed for a deeper understanding of the CNT growth and the role of the FexMn1-xO2 catalyst.
Read the original article on Max-Planck-Institut für Eisenforschung GmbH (MPIE).