Lead halide perovskites are promising semiconductors for light-emitting applications because they exhibit bright, bandgap-tunable luminescence with high colour purity. Photoluminescence quantum yields close to unity have been achieved for perovskite nanocrystals across a broad range of emission colours, and light-emitting diodes with external quantum efficiencies exceeding 20 per cent—approaching those of commercial organic light-emitting diodes—have been demonstrated in both the infrared and the green emission channels.
However, owing to the formation of lower-bandgap iodide-rich domains, efficient and colour-stable red electroluminescence from mixed-halide perovskites has not yet been realized. The report that was published in the March issue of Nature journal shows the treatment of mixed-halide perovskite nanocrystals with multidentate ligands to suppress halide segregation under electroluminescent operation.
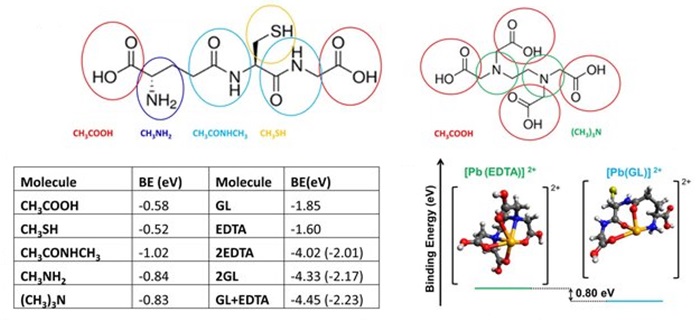
DFT-optimized structures of surface-adsorbed ligands. Structure of glutathione (left) and EDTA (right), with fragments that were used to study their interaction with the perovskite highlighted. The relative binding energies showing that EDTA binds more strongly to Pb²⁺ than does glutathione by 0.80 eV.
They demonstrate colour-stable, red emission centered at 620 nanometres, with an electroluminescence external quantum efficiency of 20.3 per cent. They show that a key function of the ligand treatment is to ‘clean’ the nanocrystal surface through the removal of lead atoms. Density functional theory calculations reveal that the binding between the ligands and the nanocrystal surface suppresses the formation of iodine Frenkel defects, which in turn inhibits halide segregation.
This work exemplifies how the functionality of metal halide perovskites is extremely sensitive to the nature of the (nano) crystalline surface and presents a route through which to control the formation and migration of surface defects. This is critical to achieve bandgap stability for light emission and could also have a broader impact on other optoelectronic applications—such as photovoltaics—for which bandgap stability is required.







