“The abuse of antibiotics has led to serious bacterial resistance, with about 1.27 million deaths in 2019 due to multidrug-resistant bacterial infections,” said co-corresponding author Linlin Li, professor at Beijing Institute of Nanoenergy and Nanosystems, Chinese Academy of Sciences. “Bacterial resistance poses a fatal threat to human health. Non-antibiotic antibacterial technologies and antibacterial nanoagents with specific catalytic activities not only produce toxic substrates to directly kill bacteria — including antibiotic-resistant bacteria — but can also reduce the risk of the bacteria developing resistance to drugs.”
According to Li, chemodynamic therapy has attracted significant attention since it was developed in 2016 as a potential companion for cancer therapies, bacterial inactivation and infectious disease treatments. The therapy uses nanomaterials containing transition metals that react with hydrogen peroxide to produce hydroxyl radical, which weakens and kills diseased or infected cells, making them more susceptible to direct treatment.
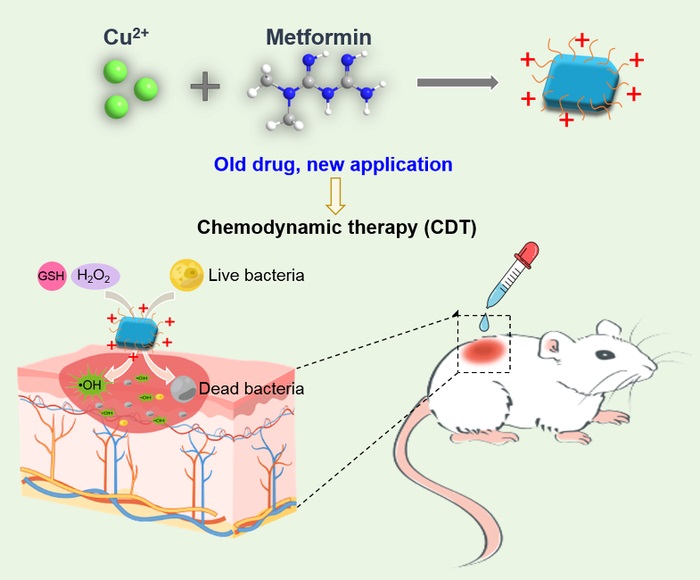
Metformin capped Cu2(OH)3Cl nanosheets are fabricated for chemodynamic antibacterial and would healing applications.
“The reaction generates toxic hydroxyl radical, which destroys the cell surface structure and causes the cell to leak its internal components,” said Li. “The nanoagents only catalyze hydroxyl radical generation at the site of tumor or inflamed tissues, so the therapy is highly specific to reduce off-target toxicity. Bacterial infections also tend to be accompanied by microenvironmental alterations that are favorable for chemodynamic therapy.”
The researchers set out to improve the antibacterial power of a nanoagent, while also lowering the toxicity to healthy cells — a risk that can be difficult to control due to the invasive nature of infection.
“Positively charged molecule parts, such as quaternary ammonium salts and chitosan, have intrinsic antibacterial effects through adsorbing on the bacteria cell wall, producing compartment resistance effect and, finally, inducing bacterial death,” said Li. “Metformin is a positively charged small molecule drug with reported anticancer, immunomodulatory and antibacterial effects. We speculated that the integration of metformin with a chemodynamic therapy nanoagent would improve the antibacterial effect.”
The researchers stirred metformin with copper chloride to form nanosheets whose surface was capped by the metformin molecules — enhancing the nanoagent’s positive charge and strengthening the antibacterial effects, according to Li.
In vitro tests revealed a biocompatible nanoplatform with a better dispersity than a nanoagent without metformin, and higher antibacterial activity.
“Compared to previous reports that used metformin as antibacterial agent alone, excellent antimicrobial effects were achieved in our experiments using lower concentrations of the nanosheets with very short action time,” Li said, who noted that they are also studying the effects of metformin in other biomedical applications to develop more therapies.
To further test the metformin-capped nanosheets, the researchers established a model of staph infection in the skin of mice. The mice were divided into five groups, each of which received a variation of treatment. The group that received both the metformin nanosheets and additional hydrogen peroxide to further the production of hydroxyl radical had the fastest wound healing rate, with complete closure by the day 12 of treatment.
“This work not only develops an efficient chemodynamic therapy nanoagent as an alternative antibacterial agent for to treat skin wound infections, but it also provides ideas about discovering new uses for old drugs,” Li said.
Read the original article on EurekAlert.