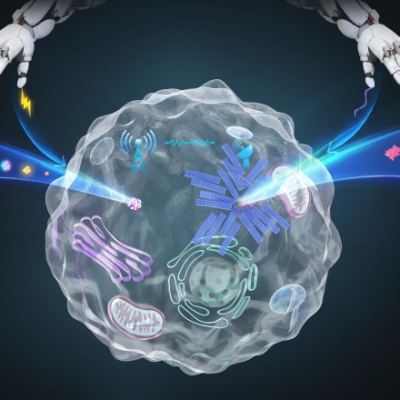
Extracellular vesicles (EVs) are naturally occurring cell-secreted lipid-bound membrane vesicles found in all biological fluids. They have gained much attention for their potential role in several biomedical applications including diagnostic purposes (biomarkers), as therapeutic agents and as drug delivery systems. The use of EVs as therapeutic agents, particularly those obtained from stem cells, has shown regenerative and/or tissue reparative effects. More importantly, EVs represent cell-free approaches as they are non-viable and work mainly via paracrine signaling, thus mitigating the challenges associated with cell-based therapy. However, several limitations need to be addressed before EVs can be extensively used as therapeutics, and these include long and tedious isolation processes, and low production yields.
In a recent paper, Associate Professor Giorgia PASTORIN and her research team from the Department of Pharmacy, National University of Singapore, in collaboration with Assistant Professor Bertrand CZARNY from Nanyang Technological University (NTU) and researchers from the Singapore Eye Research Institute (SERI), prepared nano-sized vesicles from cells via a cell shearing approach for use in wound healing and tissue regenerative applications. They found that these nano-sized vesicles called Cell-Derived Nanovesicles (CDNs) mimic the EVs not only in terms of their physical characteristics but also in their biological cargos (proteins, lipids, and nucleic acids) but can be produced at much higher yields.
Prof Pastorin said, “This research study shows that cell-derived nanovesicles hold promise for various biomedical applications. Our team has derived the nanovesicles from various cell types such as immune cells and stem cells for inflammation reduction and tissue regeneration, respectively.”
In this study, Prof Pastorin, and her doctoral student Dr Yub Raj NEUPANE in collaboration with Dr Harish K HANDRAL from the Agency for Science, Technology and Research, and Prof Czarny investigated the role that CDNs obtained from mesenchymal stem cells (MSC-CDNs) played in accelerating wound healing. As part of this investigation, high-throughput proteomics and gene ontology analysis were performed to identify the highly enriched proteins and their localisation in these vesicles. The researchers found that these MSC-CDNs activated a signaling pathway in human dermal fibroblast (HDF) cells. This increased the proliferation and migration of HDF cells towards the wound bed, along with an enhanced release of cell-proliferation related markers, growth factors and other cellular regeneration components to aid in the healing process.
This study also demonstrated that these MSC-CDNs enhanced the formation of new blood vessels in cell cultures. In murine models, they were found to promote healing at a similar extent when compared to MSC-EVs but with a healing rate that was almost twice as fast.
In future work, the team will continue exploring the use of these vesicles as therapeutic agents for cardiovascular diseases and for drug delivery purposes.

Figure showing cell-derived nanovesicles obtained from mesenchymal stem cells, which are able to accelerate would healing by mimicking natural cell-secreted extracellular vesicles.
Read the original article on National University of Singapore.