A team of scientists from Stanford University is working with researchers at the Molecular Foundry, a nanoscience user facility located at the Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab), to develop a gene-targeting, antiviral agent against COVID-19.
Last year, Stanley Qi, an assistant professor in the departments of bioengineering, and chemical and systems biology at Stanford University and his team had begun working on a technique called PAC-MAN – or Prophylactic Antiviral CRISPR in human cells – that uses the gene-editing tool CRISPR to fight influenza.
But that all changed in January, when news of the COVID-19 pandemic emerged. Qi and his team were suddenly confronted with a mysterious new virus for which no one had a clear solution. “So we thought, ‘Why don’t we try using our PAC-MAN technology to fight it?’” said Qi.
Since late March, Qi and his team have been collaborating with a group led by Michael Connolly, a principal scientific engineering associate in the Biological Nanostructures Facility at Berkeley Lab’s Molecular Foundry, to develop a system that delivers PAC-MAN into the cells of a patient.
Like all CRISPR systems, PAC-MAN is composed of an enzyme – in this case, the virus-killing enzyme Cas13 – and a strand of guide RNA, which commands Cas13 to destroy specific nucleotide sequences in the coronavirus’s genome. By scrambling the virus’s genetic code, PAC-MAN could neutralize the coronavirus and stop it from replicating inside cells.
It’s all in the delivery
Qi said that the key challenge to translating PAC-MAN from a molecular tool into an anti-COVID-19 therapy is finding an effective way to deliver it into lung cells. When SARS-CoV-2, the coronavirus that causes COVID-19, invades the lungs, the air sacs in an infected person can become inflamed and fill with fluid, hijacking a patient’s ability to breathe.
“But my lab doesn’t work on delivery methods,” he said. So on March 14, they published a preprint of their paper, and even tweeted, in the hopes of catching the eye of a potential collaborator with expertise in cellular delivery techniques.
Soon after, they learned of Connolly’s work on synthetic molecules called lipitoids at the Molecular Foundry.
Lipitoids are a type of synthetic peptide mimic known as a “peptoid” first discovered 20 years ago by Connolly’s mentor Ron Zuckermann. In the decades since, Connolly and Zuckermann have worked to develop peptoid delivery molecules such as lipitoids. And in collaboration with Molecular Foundry users, they have demonstrated lipitoids’ effectiveness in the delivery of DNA and RNA to a wide variety of cell lines.
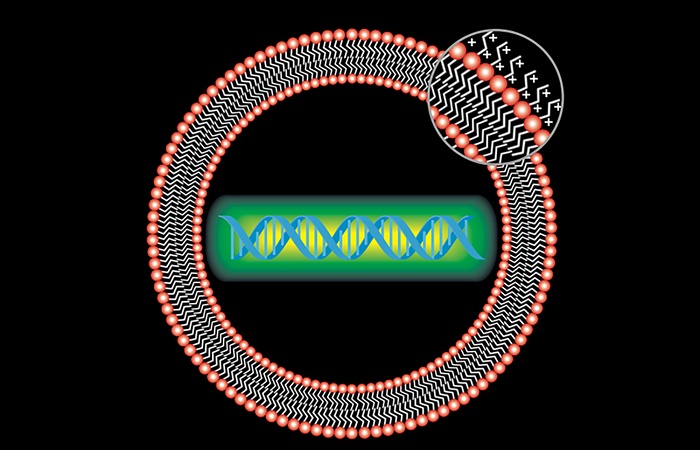
Lipitoids, which self-assemble with DNA and RNA, can serve as cellular delivery systems for antiviral therapies that could prevent COVID-19 and other coronavirus infections.
Today, researchers studying lipitoids for potential therapeutic applications have shown that these materials are nontoxic to the body and can deliver nucleotides by encapsulating them in tiny nanoparticles just one billionth of a meter wide – the size of a virus.
Now Qi hopes to add his CRISPR-based COVID-19 therapy to the Molecular Foundry’s growing body of lipitoid delivery systems.
In late April, the Stanford researchers tested a type of lipitoid – Lipitoid 1 – that self-assembles with DNA and RNA into PAC-MAN carriers in a sample of human epithelial lung cells.
According to Qi, the lipitoids performed very well. When packaged with coronavirus-targeting PAC-MAN, the system reduced the amount of synthetic SARS-CoV-2 in solution by more than 90%. “Berkeley Lab’s Molecular Foundry has provided us with a molecular treasure that transformed our research,” he said.
The team next plans to test the PAC-MAN/lipitoid system in an animal model against a live SARS-CoV-2 virus. They will be joined by collaborators at New York University and Karolinska Institute in Stockholm, Sweden.
If successful, they hope to continue working with Connolly and his team to further develop PAC-MAN/lipitoid therapies for SARS-CoV-2 and other coronaviruses, and to explore scaling up their experiments for preclinical tests.
“An effective lipitoid delivery, coupled with CRISPR targeting, could enable a very powerful strategy for fighting viral disease not only against COVID-19 but possibly against newly viral strains with pandemic potential,” said Connolly.
“Everyone has been working around the clock trying to come up with new solutions,” added Qi, whose preprint paper was recently peer-reviewed and published in the journal Cell. “It’s very rewarding to combine expertise and test new ideas across institutions in these difficult times.”
Read the original article on Berkeley Lab.