Exosomes are biological nanoparticles that are secreted into extracellular space or body fluid by almost all cell types, with a diameter of about 30 ~ 200 nm. Since exosomes exhibit a naturally derived composition and function as intercellular communication tools, they have great potential as smart vehicles for cargo delivery.
However, the commonly used methods to prepare exosomes that carry desired cargos suffer from low loading efficiency and excessive damages on both exosomes and their cargos.
In this study, Prof. YANG's group proposed a high-throughput nanofluidic device named "exosome nanoporator (ENP)", which can load a variety of exogenous cargos into exosomes.
The ENP can accurately control the characteristics of fluid in nanoscale by nanofluidic technology, so as to realize highly controllable preparation conditions of exosome-based drug delivery vehicles.
Moreover, nanofluidic channels with precise geometries were manufactured, realizing 30,000 modules working in parallel in a single device.
By transporting exosomes through the nanochannels, exosome membranes are permeabilized by mechanical compression and fluid shear, generating transient nanopores on the exosome membranes and allowing influx of cargo molecules into exosomes from surrounding solution while maintaining exosome integrity.
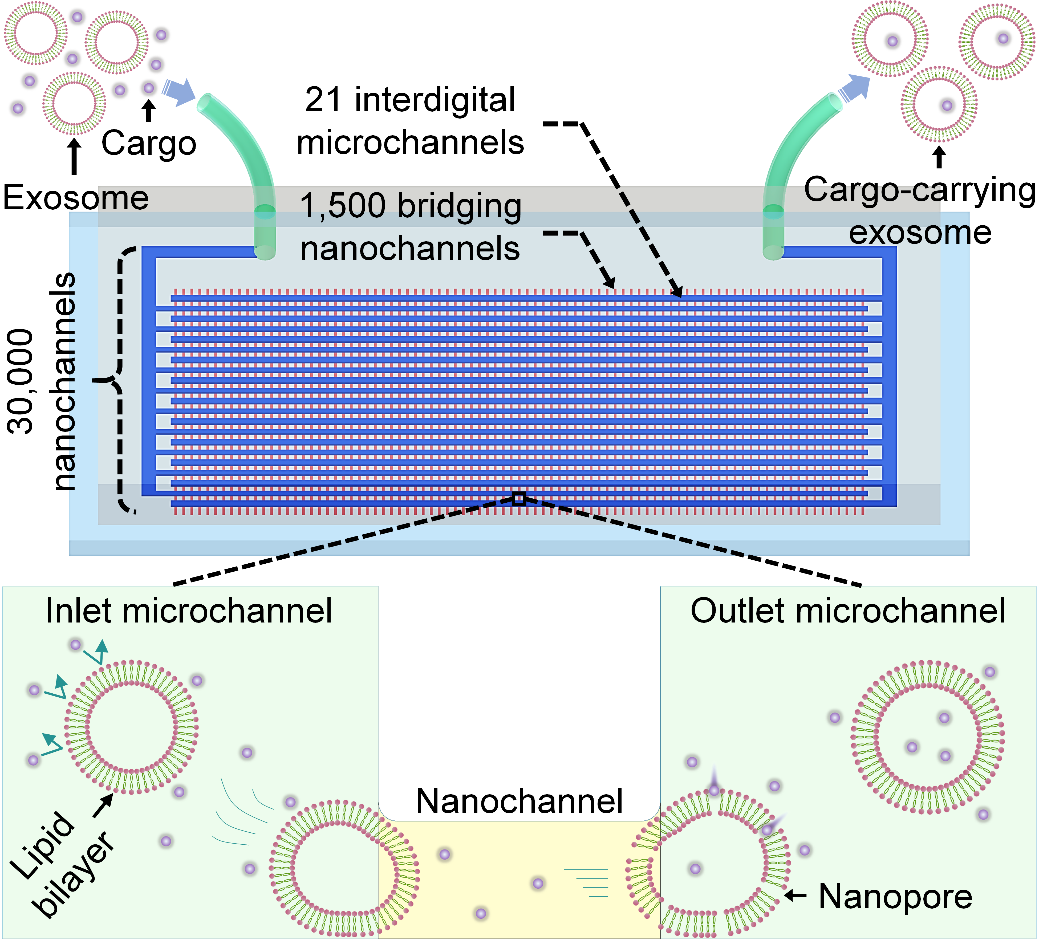
Schematic illustration of the ENP to prepare cargo-carrying exosomes.
The researchers selected a clinically approved chemotherapeutic antitumor drug, i.e. doxorubicin hydrochloride, as the cargo model, and confirmed that the ENP can efficiently load it into exosomes. "We find that the exosomes treated by the ENP can deliver their drug cargos to human non-small cell lung cancer cells and induce cell death. This indicates potential opportunities of the device for developing new exosome-based delivery vehicles for medical and biological applications," said Prof. YANG.
In the future, this new strategy is expected to develop into a platform to load different exogenous substances with biological significance and clinical therapeutic effects into exosomes. Their study was published in Small.
Read the original article on Chinese Academy of Sciences (CAS).