Jan 25, 2019
(Nanowerk News) Solar rays are a plentiful, clean source of energy that is becoming increasingly important as the world works to shift away from power sources that contribute to global warming. But current methods of harvesting solar charges are expensive and inefficient -- with a theoretical efficiency limit of 33 percent.
New nanomaterials developed by researchers at the Advanced Science Research Center (ASRC) at The Graduate Center of The City University of New York (CUNY) could provide a pathway to more efficient and potentially affordable harvesting of solar energy (Journal of Physical Chemistry C, "Singlet Fission in Combinatorial Diketopyrrolopyrrole–Rylene Supramolecular Films").
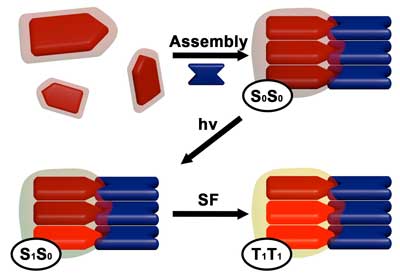 In this illustration, DPP and rylene dye molecules come together to create a self-assembled superstructure. Electrons within the structure absorb and become excited by light photons, and then couple with neighboring electrons to share energy and create additional excited electrons that can be harvested to create solar cells. (Image: Andrew Levine)
The materials, created by scientists with the ASRC's Nanoscience Initiative, use a process called singlet fission to produce and extend the life of harvestable light-generated electrons. The discovery is described in a newly published paper in the Journal of Physical Chemistry. Early research suggests these materials could create more usable charges and increase the theoretical efficiency of solar cells up to 44 percent.
"We modified some of the molecules in commonly used industrial dyes to create self-assembling materials that facilitate a greater yield of harvestable electrons and extend the electrons' xcited-state lifetimes, giving us more time to collect them in a solar cell," said Andrew Levine, lead author of the paper and a Ph.D. student at The Graduate Center.
The self-assembly process, Levine explained, causes the dye molecules to stack in a particular way. This stacking allows dyes that have absorbed solar photons to couple and share energy with -- or "excite" -- neighboring dyes. The electrons in these dyes then decouple so that they can be collected as harvestable solar energy.
In this illustration, DPP and rylene dye molecules come together to create a self-assembled superstructure. Electrons within the structure absorb and become excited by light photons, and then couple with neighboring electrons to share energy and create additional excited electrons that can be harvested to create solar cells. (Image: Andrew Levine)
The materials, created by scientists with the ASRC's Nanoscience Initiative, use a process called singlet fission to produce and extend the life of harvestable light-generated electrons. The discovery is described in a newly published paper in the Journal of Physical Chemistry. Early research suggests these materials could create more usable charges and increase the theoretical efficiency of solar cells up to 44 percent.
"We modified some of the molecules in commonly used industrial dyes to create self-assembling materials that facilitate a greater yield of harvestable electrons and extend the electrons' xcited-state lifetimes, giving us more time to collect them in a solar cell," said Andrew Levine, lead author of the paper and a Ph.D. student at The Graduate Center.
The self-assembly process, Levine explained, causes the dye molecules to stack in a particular way. This stacking allows dyes that have absorbed solar photons to couple and share energy with -- or "excite" -- neighboring dyes. The electrons in these dyes then decouple so that they can be collected as harvestable solar energy.
 In this illustration, DPP and rylene dye molecules come together to create a self-assembled superstructure. Electrons within the structure absorb and become excited by light photons, and then couple with neighboring electrons to share energy and create additional excited electrons that can be harvested to create solar cells. (Image: Andrew Levine)
The materials, created by scientists with the ASRC's Nanoscience Initiative, use a process called singlet fission to produce and extend the life of harvestable light-generated electrons. The discovery is described in a newly published paper in the Journal of Physical Chemistry. Early research suggests these materials could create more usable charges and increase the theoretical efficiency of solar cells up to 44 percent.
"We modified some of the molecules in commonly used industrial dyes to create self-assembling materials that facilitate a greater yield of harvestable electrons and extend the electrons' xcited-state lifetimes, giving us more time to collect them in a solar cell," said Andrew Levine, lead author of the paper and a Ph.D. student at The Graduate Center.
The self-assembly process, Levine explained, causes the dye molecules to stack in a particular way. This stacking allows dyes that have absorbed solar photons to couple and share energy with -- or "excite" -- neighboring dyes. The electrons in these dyes then decouple so that they can be collected as harvestable solar energy.
In this illustration, DPP and rylene dye molecules come together to create a self-assembled superstructure. Electrons within the structure absorb and become excited by light photons, and then couple with neighboring electrons to share energy and create additional excited electrons that can be harvested to create solar cells. (Image: Andrew Levine)
The materials, created by scientists with the ASRC's Nanoscience Initiative, use a process called singlet fission to produce and extend the life of harvestable light-generated electrons. The discovery is described in a newly published paper in the Journal of Physical Chemistry. Early research suggests these materials could create more usable charges and increase the theoretical efficiency of solar cells up to 44 percent.
"We modified some of the molecules in commonly used industrial dyes to create self-assembling materials that facilitate a greater yield of harvestable electrons and extend the electrons' xcited-state lifetimes, giving us more time to collect them in a solar cell," said Andrew Levine, lead author of the paper and a Ph.D. student at The Graduate Center.
The self-assembly process, Levine explained, causes the dye molecules to stack in a particular way. This stacking allows dyes that have absorbed solar photons to couple and share energy with -- or "excite" -- neighboring dyes. The electrons in these dyes then decouple so that they can be collected as harvestable solar energy.

