Sep 30, 2020
(Nanowerk News) What happens to the nanoparticles when they are injected into the bloodstream, for example, to destroy solid tumours? With new results published in ACS Nano ("Tracing the In Vivo Fate of Nanoparticles with a Non-Self Biological Identity" and "Differential Nanoparticle Sequestration by Macrophages and Scavenger Endothelial Cells Visualized in Vivo in Real-Time and at Ultrastructural Resolution"), researchers from Aarhus University are now ready to tackle such a challenging question using zebrafish embryos as a new study model in nanomedicine and nanotoxicology.
A variety of nanoparticles are designed for targeted drug delivery, but unfortunately only a very small proportion of the injected nanoparticles reach the target site such as solid tumours. The reason behind the low targeting efficiency is often considered a "black box" and had thus been little explored for many years.
Recently, an international research team led by Yuya Hayashi from the Department of Molecular Biology and Genetics (MBG), Aarhus University, demonstrated the beauty of zebrafish embryos in nano-bioimaging that can visualise dynamic interactions between nanoparticles and cells of interest in a living organism.
Now, teaming up with researchers from Interdisciplinary Nanoscience Center (iNANO), Yuya seeks to answer unsolved mysteries in bionanoscience – the first in line is the biological identity concept, which explains how cells recognise nanoparticles through a "corona" of proteins that surround each particle. This concept has now been proved for the first time in a living organism by the use of zebrafish embryos uncovering what happens to nanoparticles injected into the blood.
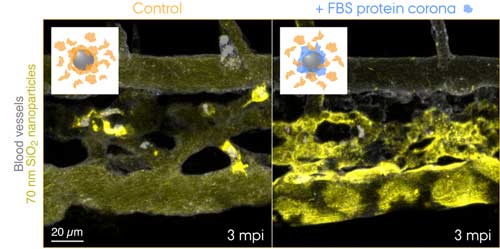 Fluorescently labelled 70 nm SiO2 nanoparticles were injected into the bloodstream of 3 days old transgenic zebrafish embryos and live-imaged at 3 min after the injection. The insets show schematics for the two conditions tested: control nanoparticles with a corona of endogenous proteins (orange) and nanoparticles with a pre-formed corona of fetal bovine serum (FBS) proteins (blue) and additional endogenous proteins. Control nanoparticles are rapidly captured by macrophages, while the nanoparticles with a non-self biological identity are effectively sequestered by scavenger endothelial cells. (Image: Yuya Hayashi. Adapted from Mohammad-Beigi et al. (2020) ACS Nano. Copyright 2020 American Chemical Society) (click on image to enlarge)
Fluorescently labelled 70 nm SiO2 nanoparticles were injected into the bloodstream of 3 days old transgenic zebrafish embryos and live-imaged at 3 min after the injection. The insets show schematics for the two conditions tested: control nanoparticles with a corona of endogenous proteins (orange) and nanoparticles with a pre-formed corona of fetal bovine serum (FBS) proteins (blue) and additional endogenous proteins. Control nanoparticles are rapidly captured by macrophages, while the nanoparticles with a non-self biological identity are effectively sequestered by scavenger endothelial cells. (Image: Yuya Hayashi. Adapted from Mohammad-Beigi et al. (2020) ACS Nano. Copyright 2020 American Chemical Society) (click on image to enlarge)
Source: By Lisbeth Heilesen, Aarhus University
Subscribe to a free copy of one of our daily
Nanowerk Newsletter Email Digests
with a compilation of all of the day's news.
 Fluorescently labelled 70 nm SiO2 nanoparticles were injected into the bloodstream of 3 days old transgenic zebrafish embryos and live-imaged at 3 min after the injection. The insets show schematics for the two conditions tested: control nanoparticles with a corona of endogenous proteins (orange) and nanoparticles with a pre-formed corona of fetal bovine serum (FBS) proteins (blue) and additional endogenous proteins. Control nanoparticles are rapidly captured by macrophages, while the nanoparticles with a non-self biological identity are effectively sequestered by scavenger endothelial cells. (Image: Yuya Hayashi. Adapted from Mohammad-Beigi et al. (2020) ACS Nano. Copyright 2020 American Chemical Society) (click on image to enlarge)
Fluorescently labelled 70 nm SiO2 nanoparticles were injected into the bloodstream of 3 days old transgenic zebrafish embryos and live-imaged at 3 min after the injection. The insets show schematics for the two conditions tested: control nanoparticles with a corona of endogenous proteins (orange) and nanoparticles with a pre-formed corona of fetal bovine serum (FBS) proteins (blue) and additional endogenous proteins. Control nanoparticles are rapidly captured by macrophages, while the nanoparticles with a non-self biological identity are effectively sequestered by scavenger endothelial cells. (Image: Yuya Hayashi. Adapted from Mohammad-Beigi et al. (2020) ACS Nano. Copyright 2020 American Chemical Society) (click on image to enlarge)

